Shuichi KOIZUMI
Dean: Faculty of Medicine, Univ Yamanashi
Professor: Dept Neuropharmacology, Univ Yamanashi
Director: Yamanashi GLIA Center
HP Link
Speciality
Glial Brain Sciences, Neuropharmacology
Research Overview
In Moonshot 7, entitled “Early Detection and Control of Dementia Onset Mechanisms Based on the Conceptual Framework Linking Glial Pathology to Senoinflammation,” I serve as a research collaborator responsible for the development of microglial transplantation and replacement technologies, thereby contributing to a fundamental understanding of brain diseases driven by chronic inflammation and to the creation of innovative intervention strategies.
In recent years, numerous studies have reported that functional alterations in glial cells play central roles in the onset and progression of a wide range of brain disorders, including dementia. However, the full molecular and cellular basis underlying these pathological processes remains insufficiently understood. In particular, microglia exhibit exceptionally high sensitivity to changes in the brain microenvironment as well as to systemic inflammatory conditions, and they undergo activation accompanied by diverse and pronounced phenotypic changes from the preclinical or early stages of disease onset, and even during the early phases of aging.
Microglia contribute not only as innate immune cells through inflammatory responses and phagocytosis, but also play essential roles in maintaining and regulating core brain functions, including synaptic remodeling and the control of neurotransmission. Therefore, functional alterations in activated microglia are considered to be deeply involved in disease initiation, chronic progression, and the development of treatment resistance (Fig. 1).
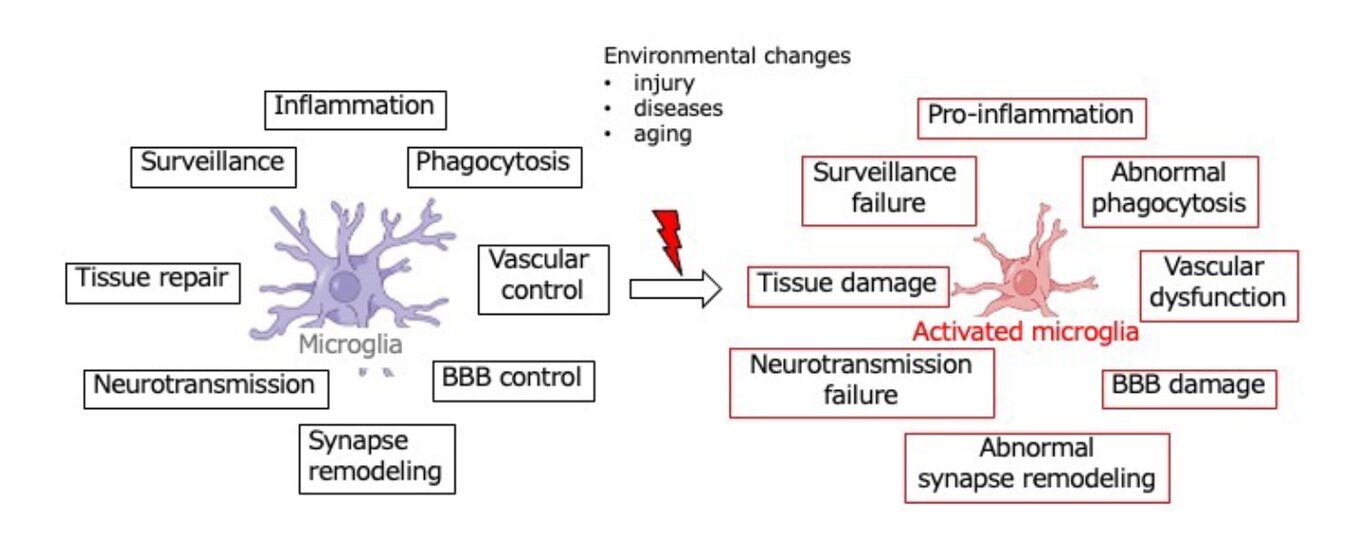
Fig.1 Diversity and alteration of microglial functions
Against this background, accurately characterizing and manipulating activated microglia represents a critically important challenge that directly links to both the understanding of brain disease pathophysiology and the establishment of innovative therapeutic strategies. As a research collaborator, I possess extensive experience and a strong research track record in glial biology and brain disease research, and have pioneered the development of a non-invasive and highly original microglial replacement technology (Parajuli et al., GLIA, 2021). This technology enables the replacement of pathologically altered microglia with normal, young, pharmacologically treated, or genetically modified microglia (Fig. 2).
By integrating this microglial replacement technology with morphological and molecular biological analyses, we aim to elucidate molecular pathologies that are either shared among or specific to different brain disorders, and to contribute substantially to the development of novel therapeutic concepts and interventions for brain diseases and age-related brain dysfunction (Fig. 3B). This approach is an original and exclusive methodology uniquely established by our group, providing a clear competitive advantage and strong originality to the present research.
Furthermore, I have been leading research on the roles of glial cells in brain and ocular diseases at both national and international levels. As the Director of the Yamanashi GLIA Center, a Center of Excellence established by the Ministry of Education, Culture, Sports, Science and Technology (MEXT) in 2021, I actively promote interdisciplinary and international research. Within Moonshot Goal 7, I am expected to play a central role in elucidating the causal relationships between glial pathology and senoinflammation.
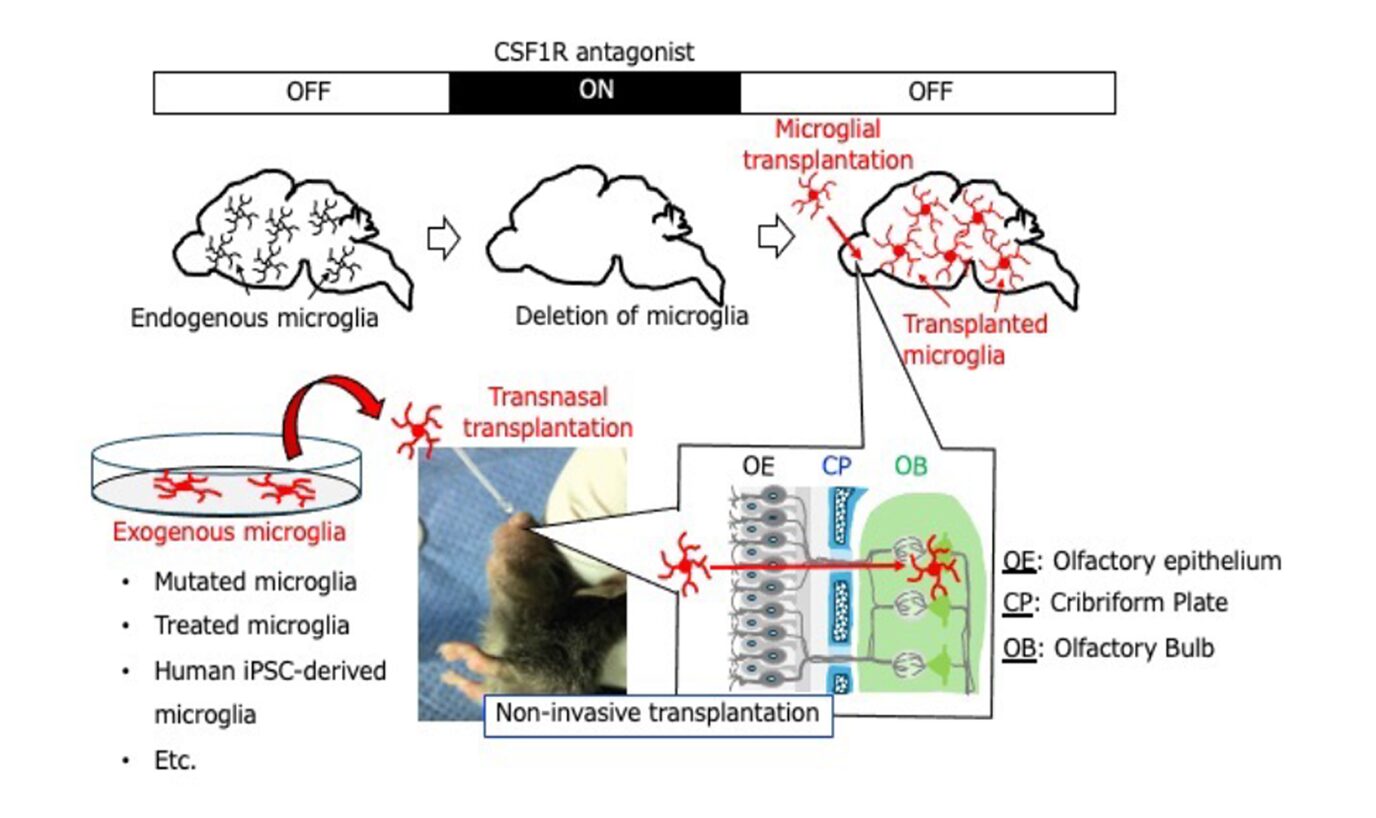
Fig. 2 Non-invasive microglial transplantation method
Main research contents
1. Elucidation of glial cell transformation patterns and glia-dependent molecular mechanisms underlying brain dysfunction in various brain diseases and aging
We have long focused on functional alterations in glial cells, which are fundamentally involved in both physiological brain functions and molecular pathology. As expressed in the statement by Miller (2005), “Everything starts with glial pathology,” many brain disorders are thought to originate from functional changes in glial cells that rapidly sense alterations in the intra- and extra-brain environment. These changes lead to subtle dysfunctions in brain activity, followed by disease onset, chronic progression, and eventual treatment resistance.
Among glial cells, microglia are exceptionally sensitive to environmental changes and undergo extensive and diverse morphological and functional transformations upon sensing such changes. In particular, microglia play a central role in senoinflammation, a pathological process in which inflammation and cellular senescence mutually reinforce each other.
Under physiological conditions, microglia perform a wide range of functions, including immune-related activities such as pro- and anti-inflammatory responses and phagocytosis, as well as the regulation of synaptic transmission and the remodeling of synapses and neural networks—processes that are directly linked to core brain functions (Fig. 1). Consequently, alterations in microglial properties and states—so-called “phenotypic transformations”—have profound effects on brain function and are causally associated with various brain diseases and age-related cognitive decline and impairment.
In this research, we aim to comprehensively analyze microglial environmental sensing mechanisms and phenotypic diversity using various disease models, from molecular biological, anatomical, and functional perspectives. Our goal is to clarify the relationships between microglial diversity, brain function, and brain diseases. As a core methodological platform, we position microglial replacement technology as a bridge from basic research to therapeutic application.
2. Development and validation of novel therapeutic strategies using non-invasive and minimally invasive microglial replacement technologies
In our previous studies, we established a completely non-invasive microglial transplantation method (intranasal transplantation) (Fig. 2). Using this approach, we demonstrated that abnormalities in microglia lead to significant impairment of brain function (Fig. 3A), and conversely, that normalization or functional enhancement of microglia promotes neuronal repair and improves brain function (Fig. 3B).
Building on these findings, the present Moonshot 7 aims to develop safer, simpler, more stable, and brain region–specific microglial transplantation and replacement technologies. By combining microglial replacement with molecular biological, anatomical, and functional analyses, we seek to elucidate the molecular mechanisms underlying brain diseases and age-related brain dysfunction. Furthermore, we aim to establish both the therapeutic efficacy and safety of microglial replacement as a novel intervention strategy.
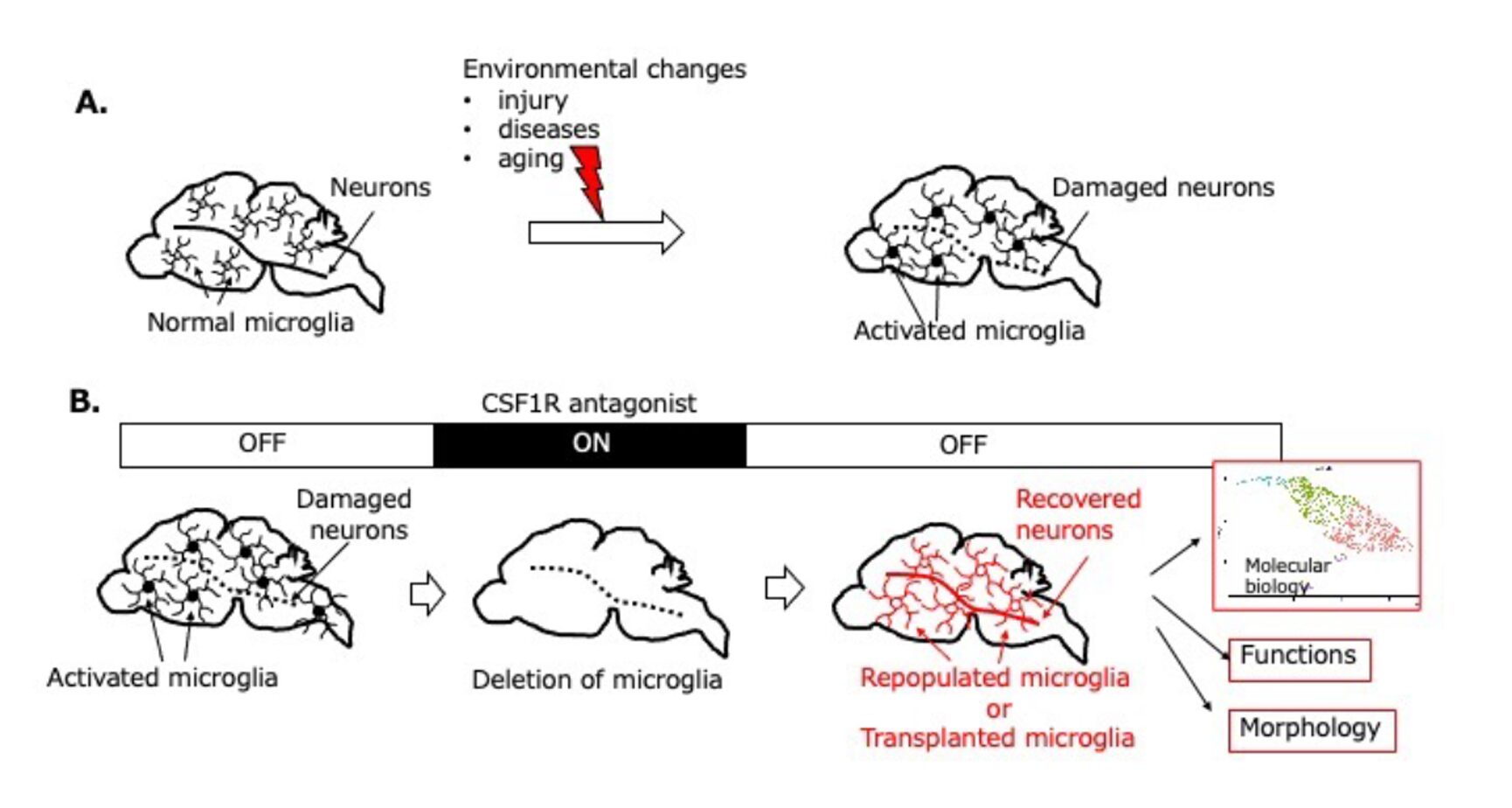
Fig.3 Development of a Novel Microglial Transplantation Strategy and Elucidation of Molecular Pathomechanisms in Neurological and Age-Related Diseases, with the Creation of Therapeutic Approaches
Major Achievements
- Kim SH, Lee J, Jang M, Roh SE, Kim S, Lee JH, Seo J, Baek J, Hwang JY, Baek IS, Lee YS, Shigetomi E, Lee CJ, Koizumi S, *Kim SK and *Kim SJ.(2025) Cerebellar Bergmann glia integrate noxious information and modulate nocifensive behaviours. Nat Neurosci, Feb;28(2):336-345.doi: 10.1038/s41593-024-01807-z.
- Shigetomi E, Suzuki H, Hirayama YJ, Sano F, Yoshihara K, Koga K, Tateoka T, Yoshioka H, Shinozaki Y, Kinouchi H, Tanaka KF, Bito H, Tsuda M and *Koizumi S. (2024) Disease-relevant upregulation of P2Y1 receptor in astrocytes enhances neuronal excitability via IGFBP2. Nat Commun 15, 6525. Doi:10.1038/s141467-024-50190-7
- Dewa K, Arimura N, Kakegawa W, Itoh M, Adachi T, Miyashita S, Hori K, Honjoya N, Yagishita H, Taya S, Miyazaki T, Usui C, Tatsumoto S, Tsuzuki A, Uetake H, Sakai K, Inoue Y, Yamakawa K, Sasaki T, Nagai J, Kawaguchi Y, Sone M, Inoue T, Go Y, Ichinohe N, Kaibuchi K, Watanabe M, Koizumi S, Yuzaki M, Hizawa K and Hoshino M. (2024) DSCAM regulates the peri-synaptic localization of GLAST in Bergmann glia for functional synapse formation. Nat Commun, 15(1) 458. doi: 10.1038/s41467-023-44579-z.
- Saito K, Shigetomi E, Shinozaki Y, Kobayashi K, Parajuli B, Kubota Y, Sakai K, Hiroshi H, Nabekura J and *Koizumi S. (2024) Microglia sense astrocyte dysfunction and prevent disease progression in an Alexander disease model. Brain, 147(2), 698-716. Doi:10.1093/brain/awad358.
- Shinozaki Y, Leung A, Namekata K, Saitoh S, Nguyen HB, Takeda A, Danjo Y, Morizawa Y, Shigetomi E, Sano F, Yoshioka N, Takebayashi H, Ohno N, Segawa T, Miyake K, Kashiwagi K, Harada T, *Ohnuma SI. and *Koizumi S. (2022) Astrocytic dysfunction induced by ABCA1 deficiency causes optic neuropathy. Sci Adv, Nov 4; 8 (44) : eabq1081. doi: 10.1126/sciadv.abq1081.
- Morizawa YM, Matsumoto M, Nakashima Y, Endo N, Aida T, Ishikane H, Beppu K, Moritoh S, Inada H, Osumi N, Shigetomi E, Koizumi S, Yang G, Hirai H, Tanaka K, Tanaka KF, Ohno N, Fukazawa Y and *Matsui K. (2022) Synaptic pruning through glial synapse engulfment during motor learning. Nat Neurosci, Nov;25(11):1458-1469. doi: 10.1038/s41593-022-01184-5.
- Takeda I, Yoshihara K, Cheung DL, Kobayashi T, Agetsuma M, Tsuda M, Eto K, Koizumi S, Wake H, Moorhouse AJ, *Nabekura J. (2022) Controlled activation of cortical astrocytes can reverse neuropathic chronic pain. Nat Commun, 13, 4100. doi:10.1038/s41467-022-31773-8
- Danjo Y, Shigetomi E, Hirayama YJ, Kobayashi K, Ishikawa T, Fukazawa Y, Shibata K, Takanashi K, Parajuli B, Shinozaki Y, Kim SK, Nabekura J and *Koizumi S. (2022) Transient astrocytic mGluR5 expression drives synaptic plasticity and subsequent chronic pain in mice. J Exp Med, April 4, 219(4), e20210989. doi: 10.1084/jem.20210989. Epub 2022 Mar 23.
- #Parajuli B, #Saito H, Shinozaki Y, Shigetomi E, Miwa H, Yoneda S, Omachi S, Asaki T, Takahashi K, Fujita M, Nakashima, K and *Koizumi S. (2021) Transnasal transplantation of human induced pluripotent stem cell-derived microglia to the brain of immunocompetent mice. GLIA, 69(10), 2332-2348. #equal contribution. doi:10.1002/glia.23985
- Saito, K.#, Shigetomi, E. #, Yasuda R., Sato R., Nakano M., Tashiro K., Tanaka, K.F., Ikenaka, K., Mikoshiba, K., Mizuta I., Yoshida,T. #, Mikoshiba, K., Nakagawa, N., Mizuno, T. and *Koizumi, S. (2018) Astrocyte aberrant Ca2+ signals “AxCa” exacerbate the pathological alterations in an Alexander disease model. GLIA. 66, 1053-1067. doi: 10.1002/glia.23300
- Morizawa YM, #Hirayama Y, #Ohno N, Shibata S, Shigetomi E, Sui Y, Nabekura J, Sato K, Okajima F, Takebayashi H, Okano H and *Koizumi S. (2017) Reactive astrocytes function as a phagocyte after brain ischemia via ABCA1-mediated pathway. Nat Commun, 8, 28. doi: 10.1038/s41467-017-00037-1
- Miyamoto, A., Wake, H., Ishikawa, A.W., Eto, K., Shibata, K., Murakoshi H, Koizumi, S., Moorhouse, A., Yoshimura, Y. and *Nabekura, J. (2016) Microglia contact induces synapse formation in developing somatosensory cortex. Nat Commun, 7, 12450. doi: 10.1038/ncomms12540.
- Kim SJ, Lee G, Bae H, Moorhouse AJ, Mikoshiba K, *Koizumi S. and *Nabekura J. (2016) Cortical astrocytes rewire somatosensory cortical circuits for peripheral neuropathic pain. J Clin Invest, 126, 1983-1997. doi: 10.1172/JCI82859
- Koizumi S, Shigemoto-Mogami Y, Nasu-Tada K, Shinozaki Y, Ohsawa K, Tsuda M, Joshi BV, Jacobson KA, Kohsaka S. and Inoue K. (2007) UDP acting at P2Y6 receptors is a novel mediator of microglial phagocytosis. Nature, 446, 1091-1095.