Makoto HIGUCHI
Director, Advanced Neuroimaging Center, Institute for Quantum Medical Science, QST
Professor, Neuroetiology and Diagnostic Science, Graduate School of Medicine, Osaka Metropolitan University
(Prospective) Director, Osaka Geroscience Institute
Link
Areas of expertise
Neuroscience, Geriatric Medicine, Diagnostic Imaging
Research Overview
As the Project Manager of Moonshot Goal 7 “Brain Senoinflammation,” I oversee the entire R&D program. I also lead the sub-project “Development and Application of Next-Generation Dedicated Brain PET Systems and Senoinflammation Imaging Technologies.” In this sub-project, our main goal is to visualize the triad of abnormal protein pathology (brain protein aging), inflammatory changes, and cellular senescence in the living brain using positron emission tomography (PET) and related technologies.
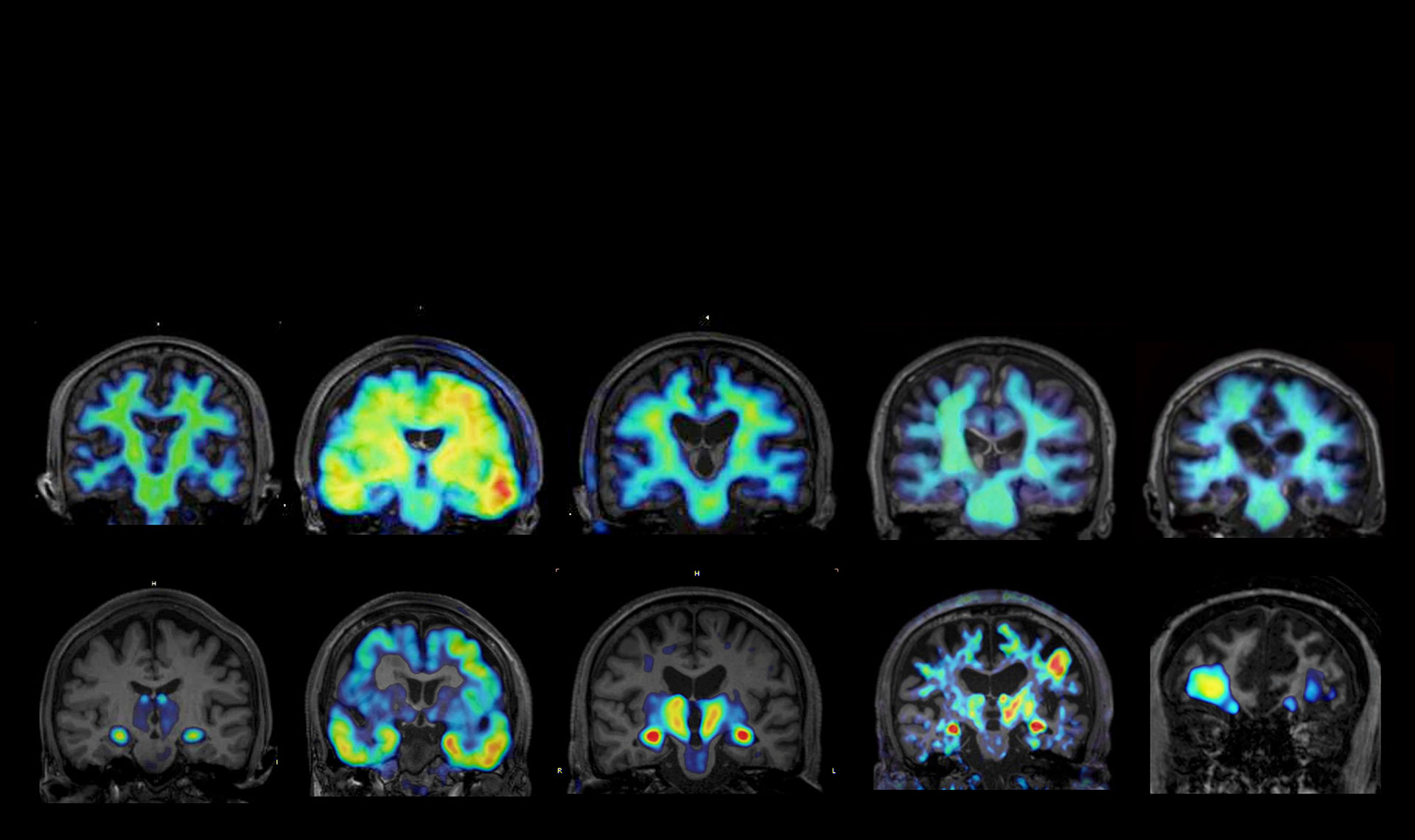
PET imaging technologies developed by QST to visualize abnormal protein pathologies in the brain cover a wide range of dementias and related disorders.

The development of the helmet-type PET system is being led in practice by Dr. Taiga Yamaya, Deputy Director at QST (https://www.qst.go.jp/site/imaging-physics/).
By establishing these imaging methods as diagnostic and evaluative tools, we will develop therapies to control this triadic pathology and promote imaging-based theranostics—treating disease while observing it through imaging. In parallel, we will accelerate the development of fluid biomarkers in blood and other body fluids, using imaging as the gold standard reference. Furthermore, to disseminate imaging as a “diagnosis anywhere” tool, we will advance the performance and mass production of a helmet-type dedicated brain PET system (VRAIN), which has been developed and implemented by QST.
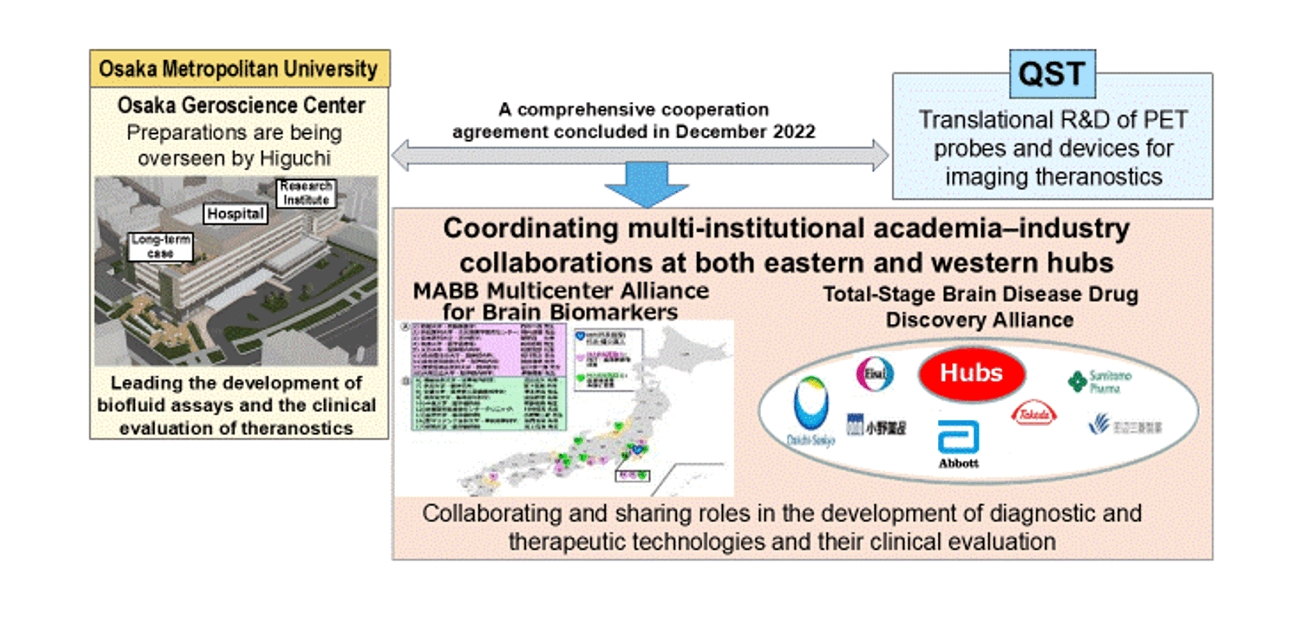
Social implementation of research accomplishments will be pursued rapidly through clinical research and trials, with QST as the eastern hub and the Osaka Geroscience Center (opening in May 2027) as the western hub. We will fully leverage the Multicenter Alliance for Brain Biomarkers (MABB) and the Total-Stage Brain Disease Drug Discovery Alliance to accelerate development.
Major Research Areas
Elucidation of Dementia Pathology and Development of Early Diagnostic and Therapeutic Strategies Based on the Concept of Senoinflammation
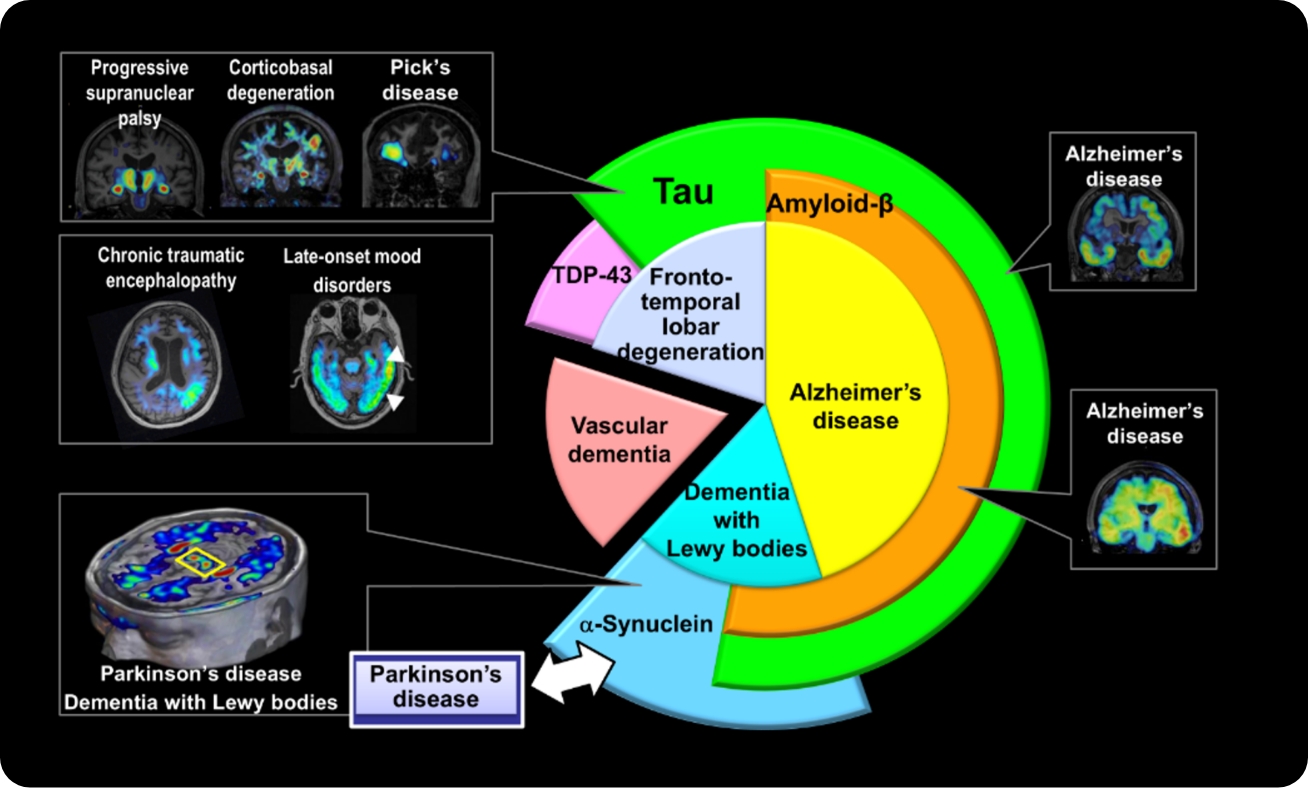
Based on our original concept that dementia pathology progresses from cerebrovascular and glial abnormalities to “senoinflammation,” we are working to elucidate disease mechanisms and establish strategies for early diagnosis and treatment. In Alzheimer’s disease and other dementias, proteins such as amyloid-β, tau, α-synuclein, and TDP-43 deviate from their normal physiological roles, undergo abnormal polymerization and aggregation, and accumulate to cause neuronal damage. These aggregates further propagate along neural circuits and expand pathology throughout the brain, contributing critically to disease progression.
We focus on “senoinflammation,” in which inflammation and cellular senescence mutually amplify each other, as the core driver of this process. We hypothesize that glial cells and cells at brain boundary regions, which normally play protective roles, undergo functional switching in response to aging or pathological stimuli and transform into “destroyers” that promote pathological protein aggregation and neurodegeneration. We are currently elucidating the molecular basis of this switching phenomenon.
Development of Novel PET Tracers to Visualize Brain Protein Aggregates and Neuroinflammation
To understand dementia pathology and enable early diagnosis, we are developing and applying novel PET tracers that visualize pathological targets in the brain. By targeting new molecules associated with protein aggregates and neuroinflammation, we aim to achieve very early diagnosis, differential diagnosis, and identification of optimal timing for therapeutic and preventive interventions.
In collaboration with pharmaceutical companies and research and medical institutions nationwide, we promote an integrated pipeline from tracer development to clinical trials and practical implementation, while also contributing to the acceleration of novel drug development based on PET imaging.
Development and Validation of Fluid Biomarkers for Dementia and Other Neurodegenerative Diseases
For very early diagnosis of dementia, the development of fluid biomarkers in blood and other body fluids is rapidly advancing worldwide. Our laboratory has pioneered highly sensitive assays to measure tau proteins and their fragments, TDP-43, and other targets in blood.
A major strength of our research is the close integration of PET tracer development and fluid biomarker studies. By using blood and cerebrospinal fluid samples from cases in which brain pathology has been rigorously identified by PET, we have a unique research platform that allows highly accurate validation of biomarkers. Leveraging this advantage, we will further promote the development and clinical validation of multi-analyte, multidimensional fluid biomarkers.
Review Article Summarizing Our Research
Higuchi M. Tagai K. Takahata K. Endo H. Advances in imaging of protein aggregates in the brain. Nature Reviews Neurology 21, 506-522, 2025. doi: 10.1038/s41582-025-01126-2
Selected Publications
Kurose S. Moriguchi S. Kubota M. Tagai K. Momota Y. Ichihashi M. Sano Y. Endo H. Hirata K. Kataoka Y. Goto R. Mashima Y. Yamamoto Y. Suzuki H. Nakajima S. Mizutani M. Sano T. Kawamura K. Zhang M.-R. Tatebe H. Tokuda T. Onaya M. Mimura M. Sahara N. Takahashi H. Uchida H. Takao M. Meyer J.H. Higuchi M. Takahata K. Diverse tau pathologies in late-life mood disorders revealed by PET and autopsy assays. Alzheimer’s & Dementia 21, e70195, 2025 doi: 10.1002/alz.70195
Tagai K. Tatebe H. Matsuura S. Hong Z. Kokubo N. Matsuoka K. Endo H. Oyama A. Hirata K. Shinotoh H. Kataoka Y. Matsumoto H. Oya M. Kurose S. Takahata K. Ichihashi M. Kubota M. Seki C. Shimada H. Takado Y. Kawamura K. Zhang MR. Soeda Y. Takashima A. Higuchi M. Tokuda T. A novel plasma p-tau181 assay as a specific biomarker of tau pathology in Alzheimer’s disease. Translational Neurodegeneration 13, 44, 2024. doi: 10.1186/s40035-024-00439-4
Endo H. Ono M. Takado Y. Matsuoka K. Takahashi M. Tagai K. Kataoka Y. Hirata K. Takahata K. Seki C. Kokubo N. Fujinaga M. Mori W. Nagai Y. Mimura K. Kumata K. Kikuchi T. Shimozawa A. Mishra SK. Yamaguchi Y. Shimizu H. Kakita A. Takuwa H. Shinotoh H. Shimada H. Kimura Y. Ichise M. Suhara T. Minamimoto T. Sahara N. Kawamura K. Zhang MR. Hasegawa M. Higuchi M. Imaging α-synuclein pathologies in animal models and patients with Parkinson’s and related diseases. Neuron 112, 2540-2557, 2024. doi: 10.1016/j.neuron.2024.05.006
Sato H, Takado Y, Toyoda S, Tsukamoto-Yasui M, Minatohara K, Takuwa H, Urushihata T, Takahashi M, Shimojo M, Ono M, Maeda J, Orihara A, Sahara N, Aoki I, Karakawa S, Isokawa M, Kawasaki N, Kawasaki M, Ueno S, Kanda M, Nishimura M, Suzuki K, Mitsui A, Nagao K, Kitamura A, Higuchi M. Neurodegenerative processes accelerated by protein malnutrition and decelerated by essential amino acids in a tauopathy mouse model. Science Advances 7, eabd5046, 2021. doi: 10.1126/sciadv.abd5046.
Tagai K, Ono M, Kubota M, Kitamura S, Takahata K, Seki C, Takado Y, Shinotoh H, Sano Y, Yamamoto Y, Matsuoka K, Takuwa H, Shimojo M, Takahashi M, Kawamura K, Kikuchi T, Okada M, Akiyama H, Suzuki H, Onaya M, Takeda T, Arai K, Arai N, Araki N, Saito Y, Trojanowski JQ, Lee VMY, Mishra SK, Yamaguchi Y, Kimura Y, Ichise M, Tomita Y, Zhang MR, Suhara T, Shigeta M, Sahara N, Higuchi M, Shimada H. High-Contrast In Vivo Imaging of Tau Pathologies in Alzheimer’s and Non-Alzheimer’s Disease Tauopathies. Neuron 109, 42-58.e8, 2021. doi: 10.1016/j.neuron.2020.09.042.
Maruyama M, Shimada H, Suhara T, Shinotoh H, Ji B, Maeda J, Zhang MR, Trojanowski JQ, Lee VM, Ono M, Masamoto K, Takano H, Sahara N, Iwata N, Okamura N, Furumoto S, Kudo Y, Chang Q, Saido TC, Takashima A, Lewis J, Jang MK, Aoki I, Ito H, Higuchi M. Imaging of tau pathology in a tauopathy mouse model and in Alzheimer patients compared to normal controls. Neuron 79, 1094-1108, 2013. doi: 10.1016/j.neuron.2013.07.037.