Naoko OHTANI
Department of Pathophysiology, Osaka Metropolitan University Graduate School of Medicine
Professor
Lab URL
Expertise
Cellular senescence, senescence-associated secretory phenotype (SASP), tumor microenvironment, gut–liver axis, gut microbiota–derived factors
Research overview
As a research collaborator in Moonshot 7, entitled “Early Detection and Control of Dementia Onset Mechanisms Based on the Conceptual Framework” evolving from glial pathology to “senoinflammation”, under the leadership of Project Manager Dr. Higuchi, I participate in this program as an expert in cellular senescence and the senescence-associated secretory phenotype (SASP).
Cellular senescence is an irreversible cell cycle arrest induced by severe cellular stress, such as extensive DNA damage that poses a risk of malignant transformation. This process functions as an intrinsic tumor-suppressive safeguard. However, accumulating evidence has revealed that senescent cells secrete a broad range of bioactive factors—including cytokines, chemokines, extracellular matrix (ECM) remodeling enzymes, growth factors, and so on —thereby profoundly altering the tissue microenvironment.
My previous work has focused primarily on the tumor microenvironment of hepatocellular carcinoma, where I have elucidated novel roles of SASP in cancer progression. These include the identification of prostaglandin E2 (PGE2) as a previously unrecognized SASP factor (Loo et al., Cancer Discovery, 2017), as well as a molecular mechanism for SASP factor release mediated by pore formation in the plasma membrane through the N-terminal domain of gasdermin D (GSDMD) (Yamagishi et al., Science Immunology, 2022).
More recently, cellular senescence and SASP have also been reported in the brain, particularly in microglia. Senescent glial cells and their SASP are increasingly implicated in “senoinflammation” in the brain, which may contribute to the onset and progression of dementia. In this project, we aim to elucidate shared molecular mechanisms of cellular senescence and SASP across multiple cell types and to clarify how these processes drive dementia pathogenesis (see Section 1 below).
In parallel, our laboratory—led by Dr. Masaki Takasugi—has conducted comprehensive proteomic analyses of organismal aging. We seek to determine whether proteins that increase or accumulate in aging organs act as molecular mediators linking systemic aging to brain aging and dementia progression (see Section 2 below). As members of this research team, Dr. Kimi Tomiyama and Dr. Maiko Uemura, both established researchers specializing in neuroscience and dementia, actively contribute to this project, enabling highly specialized, translational research aimed at dementia prevention and therapy. Within this Moonshot program, chronic inflammation is considered a key driver of dementia. Detecting, suppressing, and preventing SASP is therefore critical for the prevention and eventual overcoming of dementia. We will implement the following research proposals and further expand upon their outcomes.
Assigned Research Theme
Elucidation of the role of the senescence-associated secretory phenotype (SASP) in dementia and development of preventive strategies
1. Molecular mechanisms underlying the induction and release of SASP factors and development of preventive strategies
Although systemic chronic inflammation is suggested to contribute to dementia onset, its fundamental mechanisms remain unclear. Recent studies have demonstrated that senescent cells continuously produce inflammatory cytokines, chemokines, and other secreted factors, collectively termed the senescence-associated secretory phenotype (SASP), which contribute to chronic inflammation. Our group has previously elucidated molecular mechanisms governing the induction and release of SASP factors (Figure 1). In this project, we will utilize a p16 reporter mouse model developed by our group (Figure 2) to identify senescent organs in vivo. Based on mechanistic insights, we will develop strategies to suppress SASP induction and/or secretion. The efficacy of these interventions will be evaluated in mouse models.
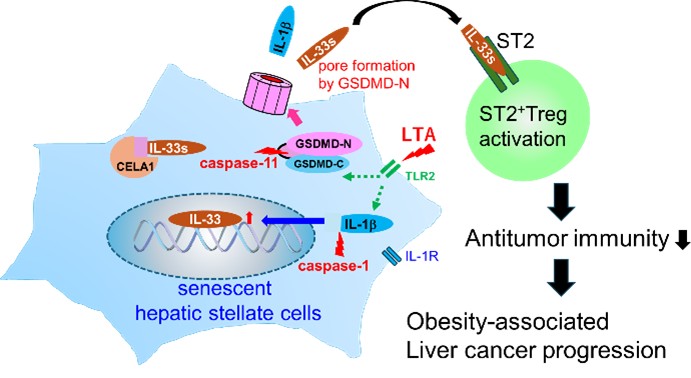
Figure 1. Release of SASP factors through pores formed in the cell membrane by the N-terminal domain of gasdermin D
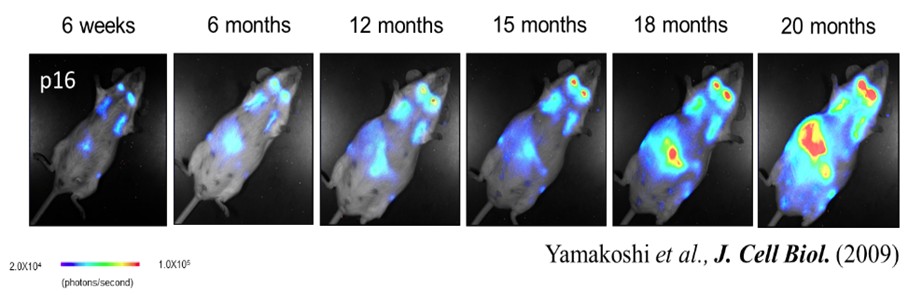
Figure 2. Real-time imaging of p16 gene expression
2. Identification of age-associated extracellular matrix factors and their interaction with SASP
Dr. Takasugi and colleagues have performed proteomic analyses to identify proteins that increase or decrease during organismal aging and have established a proteomic atlas of aging mice, including the brain. This work revealed age-associated alterations in extracellular matrix (ECM) components and the accumulation of aberrant proteins (Takasugi et al., Nature Communications, 2024). In this project, we will identify ECM proteins that change with aging and investigate their functional interactions with SASP factors. Through targeted removal or degradation of accumulated proteins, we aim to clarify the relationship between organismal aging-associated protein accumulation and SASP. Furthermore, we will examine not only individual aging organs but also inter-organ communication and its impact on the brain microenvironment, ultimately contributing to the development of preventive strategies for dementia.
3. Development of therapeutic strategies to suppress or reverse aberrant microglia and senescent neurons in aging and dementia
Drs. Tomiyama and Umeda have demonstrated that aberrant microglia appearing in dementia express gpNMB, and that administration of anti-gpNMB antibodies normalizes microglial function and improves cognitive performance in animal models. In this project, we will elucidate how gpNMB contributes to cellular senescence and chronic inflammation, as well as its effects on the surrounding microenvironment. We aim to clarify the mechanism by which anti-gpNMB antibodies restore microglial homeostasis. In addition, we are developing food-derived compounds and supplements capable of suppressing senescent cells (p16-, p21-, and γH2AX-positive neurons) in the aged mouse brain, rewinding the brain epigenetic clock, and restoring age-associated cognitive decline, with the goal of dementia prevention and treatment.
Lab HP: https://www.omu.ac.jp/med/neurosci/
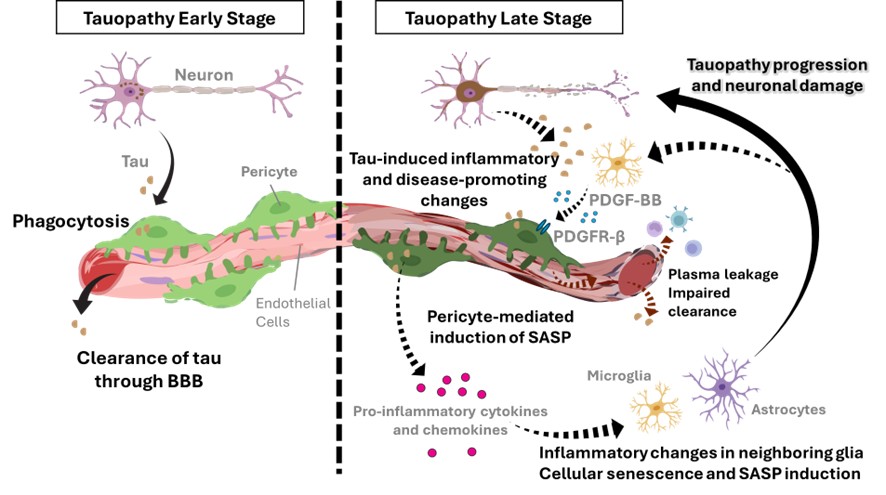
4. Control of tauopathy progression through identification of pathology-associated pericytes
Dr. Maiko Uemura and colleagues have demonstrated in a tau proteinopathy mouse model of dementia that cellular senescence in the brain becomes prominent as the pathology progresses, accompanied by the induction of SASP in both vascular and glial cell lineages. Focusing on pericytes, which reside in the perivascular niche and function as a cellular link between these two systems, they found that genetic partial deletion of the pericyte-characteristic platelet-derived growth factor receptor beta (PDGFRβ) suppresses perivascular extracellular matrix accumulation, inflammatory changes in glial cells, neuronal degeneration, and tau protein accumulation. Notably, these findings indicate that pericytes, which normally act as ‘guardians’ maintaining perivascular homeostasis, undergo a functional conversion into inflammatory, disease-promoting ‘disruptors.’ Building on this work, the present project aims to elucidate the mechanisms underlying pericyte transformation and to develop diagnostic biomarker assays and novel therapeutic strategies targeting pericyte-associated molecules.
Lab HP: https://www.omu.ac.jp/med/research/departments/etiology-diagnostic-sciences/index.html
Research Achievements of the Participating Researcher
- Takasugi M, Nonaka Y, Takemura K, Yoshida Y, Stein F, Schwarz J.J, Adachi J, Satoh J, Ito S, Tombline G, Biashad S.A, Seluanov A, Gorbunova V, Ohtani, N. An atlas of the aging mouse proteome reveals the features of age-related post-transcriptional dysregulation. Nat Commun 2024 15(1):8520
- Kawamoto S, Uemura K, Hori N, Takayasu L, Konishi Y, Katoh K, Matsumoto T, Suzuki M, Sakai Y, Matsudaira T, Adachi T, Ohtani N, Standley DM, Suda W, Fukuda S, Hara E. Bacterial induction of B cell senescence promotes age-related changes in the gut microbiota. Nat Cell Biol. 2023 Jun;25(6):865-876.
- Yamagishi R, Kamachi F, Nakamura M, Yamazaki S, Kamiya T, Takasugi M, Cheng Y, Nonaka Y, Yukawa-Muto Y, Thuy LTT, Harada Y, Arai T, Loo TM, Yoshimoto S, Ando T, Nakajima M, Taguchi H, Ishikawa T, Akiba H, Miyake S, Kubo M, Iwakura Y, Fukuda S, Chen WY, Kawada N, Rudensky A, Nakae S, Hara E, Ohtani N.Gasdermin D-mediated release of IL-33 from senescent hepatic stellate cells promotes obesity-associated hepatocellular carcinoma. Science Immunol. 2022 Jun 24;7(72):eabl7209.
- Takahashi A, Loo TM, Okada R, Kamachi F, Watanabe Y, Wakita M, Watanabe S, Kawamoto S, Miyata K, Barber GN, Ohtani N, Hara E. Downregulation of cytoplasmic DNases is implicated in cytoplasmic DNA accumulation and SASP in senescent cells. Nat Commun. 2018 Mar 28;9(1):1249. doi: 10.1038/s41467-018-03555-8.
- Loo TM, Kamachi F, Watanabe Y, Yoshimoto S, Kanda H, Arai Y, Nakajima-Takagi Y, Iwama A, Koga T, Sugimoto Y, Ozawa T, Nakamura M, Kumagai M, Watashi K, Taketo MM, Aoki T, Narumiya S, Oshima M, Arita M, Hara E, Ohtani N. Gut Microbiota Promotes Obesity-Associated Liver Cancer through PGE2-Mediated Suppression of Antitumor Immunity. Cancer Discov. 2017 May;7(5):522-538.
- Yoshimoto S, Loo TM, Atarashi K, Kanda H, Sato S, Oyadomari S, Iwakura Y, Oshima K, Morita H, Hattori M, Honda K, Ishikawa Y, Hara E, Ohtani N. Obesity-induced gut microbial metabolite promotes liver cancer through senescence secretome. Nature. 2013 Jul 4;499(7456):97-101.
- Umeda T, Sakai A, Uekado R, Shigemori K, Nakajima R, Yamana K, Tomiyama T. Simply crushed zizyphi spinosi semen prevents neurodegenerative diseases and reverses age-related cognitive decline in mice. Elife. 2025 Apr 23;13:RP100737. doi: 10.7554/eLife.100737.
- Umeda T, Sakai A, Shigemori K, Nakata K, Nakajima R, Yamana K, Tomiyama T. New Value of Acorus tatarinowii/gramineus Leaves as a Dietary Source for Dementia Prevention. Nutrients. 2024 May 23;16(11):1589. doi: 10.3390/nu16111589.
- Umeda T, Shigemori K, Uekado R, Matsuda K, Tomiyama T. Hawaiian native herb Mamaki prevents dementia by ameliorating neuropathology and repairing neurons in four different mouse models of neurodegenerative diseases. Geroscience. 2024 Apr;46(2):1971-1987. doi: 10.1007/s11357-023-00950-y.
- Uemura MT, Suh ER, Robinson JL, Brunden KR, Grossman M, Irwin DJ, Lee VM, Trojanowski JQ, Lee EB, Van Deerlin VM. Abundant co-pathologies of polyglucosan bodies, frontotemporal lobar degeneration with TDP-43 inclusions, and ageing-related tau astrogliopathy in a family with a GBE1 mutation. Neuropathol Appl Neurobiol. 2022 Dec 1:e12865. doi: 10.1111/nan.12865.
- Uemura MT, Robinson JL, Cousins KAQ, Tropea TF, Kargilis DC, McBride JD, Suh E, Xie SX, Xu Y, Porta S, Uemura N, Van Deerlin VM, Wolk DA, Irwin DJ, Brunden KR, Lee VM, Lee EB, Trojanowski JQ. Distinct characteristics of limbic-predominant age-related TDP-43 encephalopathy in Lewy body disease. Acta Neuropathol. 2022 Jan;143(1):15-31. doi: 10.1007/s00401-021-02383-3.
- Taguchi T, Ikuno M, Hondo M, Parajuli LK, Taguchi K, Ueda J, Sawamura M, Okuda S, Nakanishi E, Hara J, Uemura N, Hatanaka Y, Ayaki T, Matsuzawa S, Tanaka M, El-Agnaf OMA, Koike M, Yanagisawa M, Uemura MT, Yamakado H, Takahashi R. “α-Synuclein BAC transgenic mice exhibit RBD-like behaviour and hyposmia: a prodromal Parkinson’s disease model.” Brain.
- Uemura MT, Ihara M, Maki T, Nakagomi T, Kaji S, Uemura K, Matsuyama T, Kalaria RN, Kinoshita A, Takahashi R. “Pericyte-derived Bone Morphogenetic Protein 4 Underlies White Matter Damage after Chronic Hypoperfusion.” Brain Pathology, 2018