Mitsutoshi SETOU
Professor, Department of Cellular and Molecular Anatomy, Faculty of Medicine, Hamamatsu University School of Medicine
Director, International Mass Imaging and Spatial Omics Center, Hamamatsu University School of Medicine
Laboratory Links
Field of Expertise
Anatomy, Medical Mass Spectrometry, Quantum Life Sciencce, Aging and Age-related Diseases, Post-translational Modification
Research Overview
We lead the We lead the assigned research sub-theme “Development of disease prevention through cellular rejuvenation using glia-targeting drugs and blockade of aging-promoting factors.”
With mass spectrometry as our core technological platform, we pursue research aimed at elucidating the molecular mechanisms underlying the onset and progression of neurodegenerative diseases, and at translating these insights into innovative diagnostic and therapeutic strategies. Within the Moonshot Research and Development Program, our role is to precisely capture, at the molecular level, disease states in which aberrant protein pathology, neuroinflammation, and cellular senescence dynamically interact in the brain.
In neurodegenerative disorders such as Alzheimer’s disease, Parkinson’s disease, and multiple system atrophy, pathological proteins—including tau and α-synuclein—misfold, aggregate, and propagate through neural circuits, thereby driving disease progression. A defining feature of our research is the direct identification and analysis of these pathological processes by mass spectrometry, without relying on predefined markers or hypothesis-driven assumptions.
Our research outcomes are expected to contribute not only to the development of ultra-early diagnostic biomarkers for neurodegenerative diseases, but also to the creation of disease-modifying therapeutics based on the regulation of microglial function. By bridging mass spectrometry–based molecular science with drug discovery research, we aim to advance next-generation therapeutic strategies for neurodegenerative disorders.
Main Research Topics
Based on molecular entity information obtained through mass spectrometry, we integrate our research with PET-based brain imaging and pharmacological efficacy studies. This multidisciplinary approach enables comprehensive evaluation of how candidate compounds influence microglial functions and disease-related molecular networks, thereby promoting drug discovery research that unifies diagnosis and therapy.
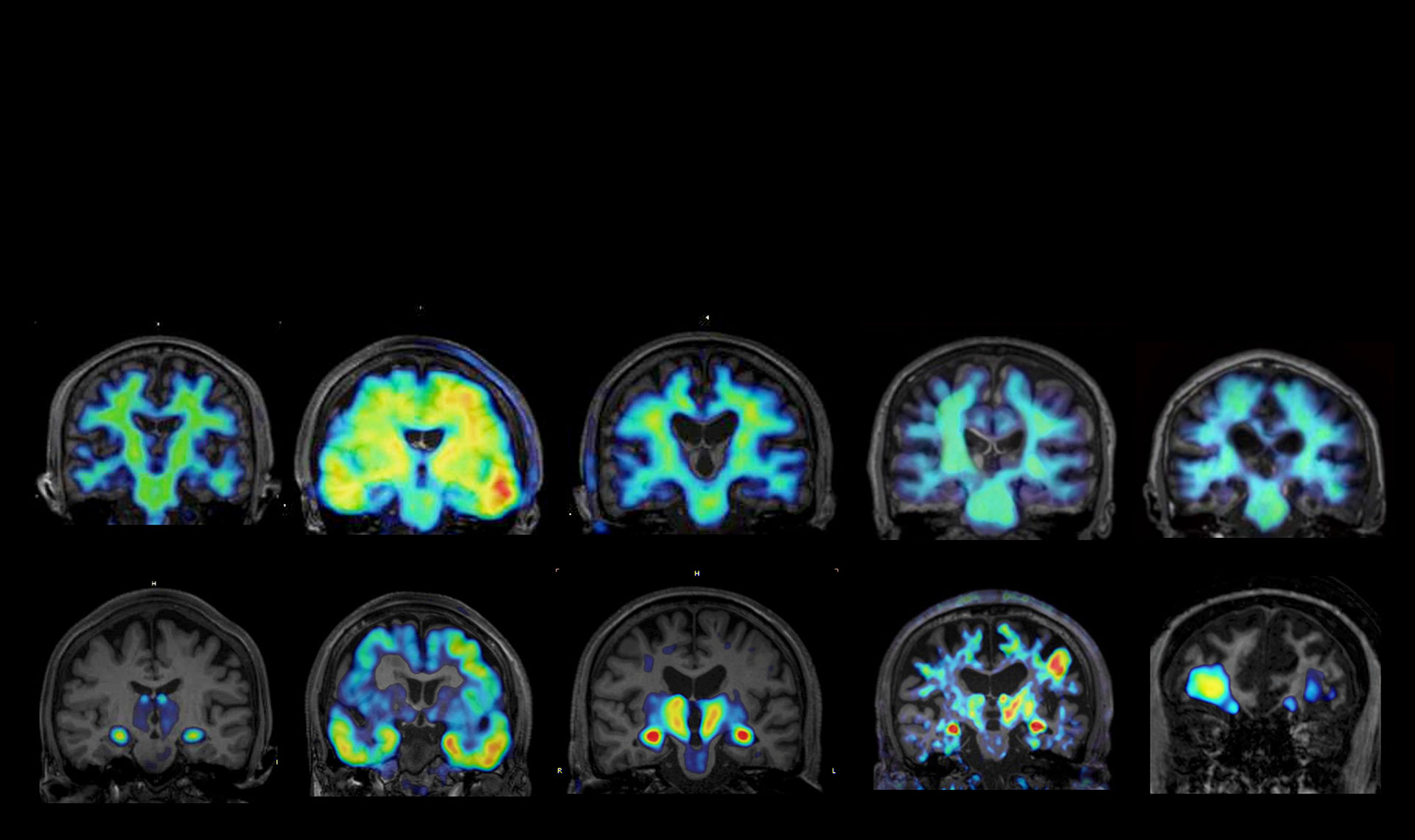
Spatial molecular mapping of disease pathology using mass spectrometry imaging
We employ mass spectrometry imaging techniques (including MALDI and DESI) to visualize, at high spatial resolution, the distributions of proteins, peptides, and lipids within brain tissue. This approach allows us to identify disease-associated molecules and molecular networks formed in and around pathological lesions that cannot be fully captured by conventional immunohistochemistry.
Molecular characterization of pathological protein aggregates and identification of therapeutic targets
Through comprehensive proteomic and protein–protein interaction analyses, we characterize the molecular composition and post-translational modifications of pathological protein aggregates. These analyses enable the identification of disease-specific molecular species and pathogenic molecular networks, providing a robust foundation for therapeutic target discovery.
Mechanistic analysis of microglial recognition and clearance of pathological proteins
Alterations in glial cell function, particularly in microglia, play a central role in the progression of neurodegenerative diseases. We investigate how microglia recognize, internalize, degrade, or inadvertently propagate pathological proteins using mass spectrometry–based molecular profiling.
UBL3-mediated membrane trafficking and exosome pathways as a platform for drug discovery
We focus on the ubiquitin-like protein UBL3 and have elucidated its role in regulating protein dynamics through membrane trafficking and extracellular vesicle (exosome) pathways. By quantitatively assessing, using mass spectrometry, the extracellular release of pathological proteins and their subsequent uptake and degradation by microglia, we are establishing novel drug discovery concepts aimed at controlling disease progression.
Research Papers
Yan J, Kahyo T, Zhang H, Ping Y, Zhang C, Jiang S, Ji Q, Ferdous R, Islam MS, Oyama S, Aramaki S, Sato T, Mimi MA, Hasan MM, Setou M. Alpha-synuclein interaction with UBL3 is upregulated by microsomal glutathione S-transferase 3, leading to increased extracellular transport of the alpha-synuclein under oxidative stress. Int J Mol Sci 2024 25(13):7353. doi: 10.3390/ijms25137353
Yan J, Zhang H, Tomochika Y, Chen B, Ping Y, Islam MS, Aramaki S, Sato T, Nagashima Y, Nakamura T, Kahyo T, Kaneda D, Ogawa K, Yoshida M, Setou M. UBL3 interaction with α-synuclein is downregulated by silencing MGST3. Biomedicines 2023,11(9):2491. doi: 10.3390/biomedicines11092491
Chen B, Hasan MM, Zhang H, Zhai Q, Waliullah ASM, Ping Y, Zhang C, Oyama S, Mimi MA, Tomochika Y, Nagashima Y, Nakamura T, Kahyo T, Ogawa K, Kaneda D, Yoshida M, Setou M. UBL3 interacts with α-synuclein in cells and the interaction is downregulated by the EGFR pathway inhibitor Osimertinib. Biomedicines 2023,11(6):1685. doi: 10.3390/biomedicines11061685
Ageta H, Ageta-Ishihara N, Hitachi K, Karayel O, Onouchi T, Yamaguchi H, Kahyo T, Hatanaka K, Ikegami K, Yoshioka Y, Nakamura K, Kosaka N, Nakatani M, Uezumi A, Ide T, Tsutsumi Y, Sugimura H, Kinoshita M, Ochiya T, Mann M, Setou M, Tsuchida K, UBL3 modification influences protein sorting to small extracellular vesicles. Nat Commun 2018, 9(1), 3936. doi: 10.3390/neurolint16060089
Phua SC, Chiba S, Suzuki M, Su E, Elle RC, Ganesh PV, Setou M, Rohatgi R, Jeremy RF, Ikegami K, Inoue T. Dynamic remodeling of membrane composition drives cell cycle through primary cilia excision. Cell 2017, 168:1-2:264-279. doi: 10.1016/j.cell.2016.12.032