Ayuko HOSHINO
Research Center for Advanced Science and Technology, the University of Tokyo
Intercellular Communication & Medical Science Professor
Graduate School of Life Sciences, Tohoku University Professor
InaRIS Fellow
ST-AR Project Representative Director
Links
field of expertise
Exosome, Autism Spectrum Disorder (ASD), Pregnancy Complications, Cancer, Metastasis, Disease Biology, Inter-organ Communication
Overview of Our Research
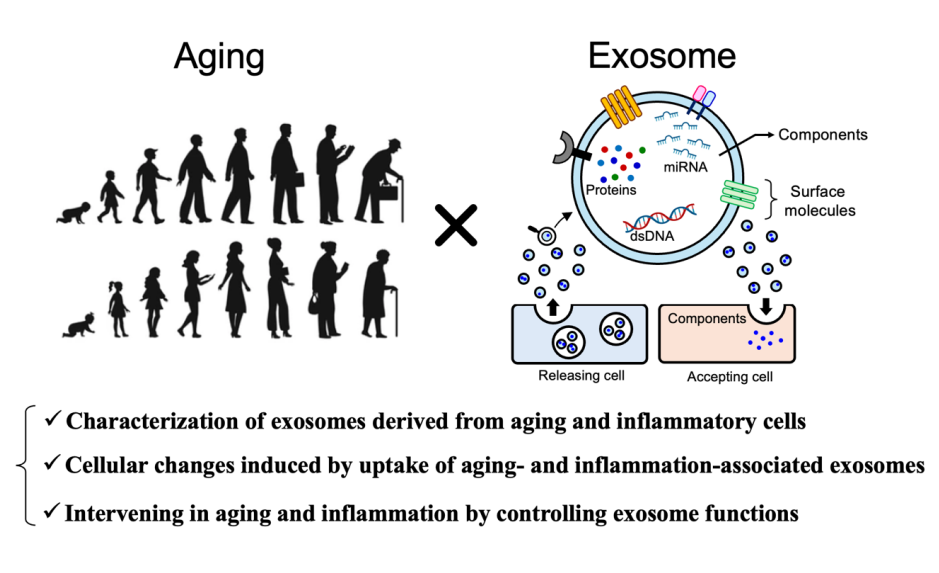
Our research aims to uncover what happens in the body during aging and chronic inflammation, with a particular focus on how the brain environment changes. To achieve this, we focus on exosomes, tiny particles that mediate communication between cells.
Exosomes are nanosized vesicles released by cells that carry information molecules such as proteins and microRNAs (miRNAs). They circulate throughout the body via bodily fluids, including the blood, and can reach organs such as the brain. Recent studies have shown that exosomes released from aged or inflamed cells can alter the properties and functions of other cells. However, it remains largely unknown what kinds of information these exosomes carry, which cells take them up, and what changes they induce.
In this project, we systematically investigate the identity, destinations, and effects of aging- and inflammation-associated exosomes. While placing a central focus on glial cells in the brain, we also examine immune cells and peripheral organs. Through this comprehensive approach, we aim to deepen our understanding of senoinflammation in the brain and pave the way for the development of novel intervention strategies.
Theme 1: Characterization of Exosomes Derived from Aged and Inflammatory Cells
Cells undergoing aging or chronic inflammation are thought to release exosomes with properties distinct from those produced under healthy conditions. In this theme, we analyze exosomes circulating in the blood as well as those produced by the brain, immune cells, and other peripheral organs, focusing on:
・Their size and abundance
・The types of proteins they contain
・miRNAs that regulate gene expression
By doing so, we aim to identify aging- and inflammation-specific exosomal “fingerprints” (signatures) and determine which organs and cell types play central roles in these changes. In the brain in particular, we go beyond morphological alterations and focus on differences in exosomal cargo, allowing us to capture brain-specific mechanisms of information transfer associated with aging and inflammation.
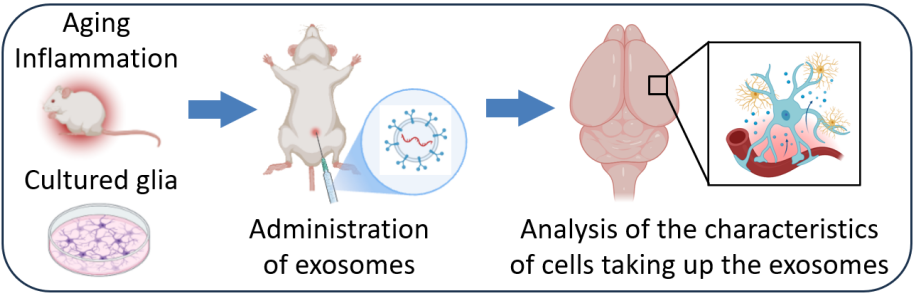
Theme 2: Cellular Changes Induced by Uptake of Aging- and Inflammation-Related Exosomes
For exosomes to exert their effects, they must first be taken up by recipient cells. In this theme, we investigate what happens inside cells after they receive aging- or inflammation-associated exosomes.
In the brain, led by Dr. Yutaro Kashiwagi, we focus on glial cells—including astrocytes and microglia—which support neuronal function and play key roles in immune and inflammatory responses. Specifically, we examine:
・Which cell types preferentially take up aging- and inflammation-related exosomes
・How gene expression changes after exosome uptake
・How cellular morphology and cell–cell interactions are altered
By combining super-resolution microscopy with gene expression analyses, we aim to understand how senoinflammation progresses in the brain at the cellular level.
Importantly, this analysis is extended beyond the brain to include immune cells and cells from other organs, enabling a comprehensive understanding of how aging- and inflammation-related exosomes act throughout the body. By identifying key molecules involved in exosome uptake, we also seek to lay the foundation for future therapeutic interventions.
Theme 3: Analysis of Exosomes Released from Aged and Inflammatory Glial Cells
In this theme, we focus on glial cells as a potential source of senoinflammation in the brain. Glial cells not only support neurons but also play essential roles in brain immunity and inflammatory responses, and they are known to be strongly affected by aging and chronic inflammation.
We hypothesize that glial cells in aged or inflammatory states release exosomes with properties distinct from those under normal conditions. To test this, we compare exosomes isolated from the brains of aged and young mice, and we also analyze exosomes produced by cultured glial cells under aging- or inflammation-mimicking conditions.
Specifically, we examine exosomes released from multiple glial cell types—astrocytes, microglia, and oligodendrocytes—focusing on differences in size, abundance, and protein and miRNA content. This approach allows us to identify glial-derived exosomal signatures that are specific to aging and inflammation.
Furthermore, we analyze how these glial-derived exosomes affect recipient cells, including neighboring neurons, other glial cells, and even cells outside the brain. By elucidating the interactions between glial cells as information senders and recipient cells as information receivers, we aim to clarify how senoinflammation in the brain is maintained and amplified over time.
References
- Yamagata C, Hamazaki Y, Nakazato T, Itai S, Honjo M, Kato M, Kurashina Y, Asai M, Hoshino A*, Onoe H*, “Oil-sealed RGD-modified alginate hydrogel microwell array for analysis of single-cell-derived extracellular vesicles and particles,” Microsystems & Nanoengineering 2025, Dec 2;11(1):238. doi: 10.1038/s41378-025-01092-1 (co-corresponding author)
- Hirosawa K, Sato Y, Kasai R, Yamaguchi E, Komura N, Ando H, Hoshino A, Yokota Y & Suzuki K. Uptake of small extracellular vesicles by recipient cells is facilitated by paracrine adhesion signaling. Nature Communications 2025, 16, 2419
- Bojmar L, Kim HS, Sugiura K, Heissel S, Lucotti S, Cioffi M, Johnson KE, Cohen-Gould L, Zhang H, Molina H, Matei IR, Lyden D, Hoshino A. Protocol for cross-platform characterization of human. STAR Protocols 2023, 5(1)102754
- Kenari AN, Bojmar L, Heissel S, Molina H, Lyden D, Hoshino A. Protocol for Plasma Extracellular Vesicle and Particle Isolation and Mass Spectrometry-Based Proteomic Identification. Methods Mol Biol. 2023 ;2628:291-300. doi: 10.1007/978-1-0716-2978-9_19. PMID: 36781793.
- Hashimoto A, Sugiura K, Hoshino A. Impact of exosome-mediated feto-maternal interactions on pregnancy maintenance and development of obstetric complications. The Journal of Biochemistry 2021 Mar 5;169(2):163-171
- Hoshino A, Sang Kim H, Bojmar L, Ennu Gyan K, Cioffi M, Hernandez J, Zambirinis CP, Rodrigues G, Molina H, Heissel S, Tesic Mark M, Steiner L, Benito-Martin A, Lucotti S, Di Giannatale A, Offer K, Nakajima M, Williams C, Nogués L, Pelissier Vatter FA, Hashimoto A, Davies AE, Freitas D, Kenific C, Ararso Y, Buehring W, Lauritzen P, Ogitani Y, Sugiura K, Takahashi N, Aleckovic M, Bailey KA, Jolissant JS, Wang H, Harris A, Schaeffer LM, Posner Z, Balachandran VP, Khakoo Y, Raju P, Scherz A, Sagi I, Scherz-Shouval R, Yarden Y, Oren M, Petriccione M, De Braganca K, Donzelli M, Fischer C, Vitolano S, Wright G, Ganshaw L, Marrano M, Ahmed A, DeStefano J, Danzer E, Roehrl MH, Lacayo NJ, Vincent T, Weiser MR, Brady MS, Meyers P, Wexler LH, Ambati S, Chou AJ, Slotkin E, Modak S, Roberts S, Basu E, Diolaiti D, Krantz B, Cardoso F, Simpson AL, Berger M, Rudin CM, Simeone DM, Jain M, Ghajar CM, Batra SK, Stanger BZ, Bui J, Brown KA, Rajasekhar VK, Healey JH, de Sousa M, Kramer K, Sheth S, Baisch J, Pascual V, Heaton TE, La Quaglia MP, Pisapia DJ, Schwartz R, Zhang H, Liu Y, Shukla A, Sarte L, DeClerck Y, LaBarge M, Bissell MJ, Grandgenett P, Hollingsworth M, Bromberg J, Costa-Silva B, Peinado H, Kang Y, Garcia BA, O’Reilly E, Kelsen D, Trippett TM, Jones DR, Matei I, Jarnagin WR, Lyden D. Extracellular Vesicle and Particle biomarkers define multiple human cancers. Cell 2020 Aug 20;182(4):1044-1061 (First and Corresponding author).
- Rodrigues G*, Hoshino A*, Matei IR, Scandariato I, Kenific CM, Kim HS, Casanova-Salas I, Dai D, Badwe CR, Gril B, Mark MT, Dill BD, Molina H, Benito-Martin A, Bojmar L, Offer K, LaPlant Q, Ararso Y, Buehring W, Wang H, Juan X, Liu Y, Sabari JK, Shin SJ, Narula N, Rajasekhar VK, Zhang H, Costa-Silva B, Rafii S, Rudin CM, Jones DR, Steeg PS, Peinado H, Ghajar CM, Bromberg J, Pisapia D, de Sousa M, Lyden D. Tumour exosomal CEMIP protein promotes cancer cell colonization in brain metastasis. Nature Cell Biology 2019 Nov 4 (Co-first author)
- Hoshino A, Costa-Silva B, Shen TL, Rodrigues G, Hashimoto A, Tesic Mark M, Molina H, Kohsaka S, Di Giannatale A, Ceder S, Singh S, Williams C, Soplop N, Uryu K, Pharmer L, King T, Bojmar L, Davies AE, Ararso Y, Zhang T, Zhang H, Hernandez J, Weiss JM, Dumont-Cole VD, Kramer K, Wexler LH, Narendran A, Schwartz GK, Healey JH, Sandstrom P, Jorgen Labori K, Kure EH, Grandgenett PM, Hollingsworth MA, de Sousa M, Kaur S, Jain M, Mallya K, Batra SK, Jarnagin WR, Brady MS, Fodstad O, Muller V, Pantel K, Minn AJ, Bissell MJ, Garcia BA, Kang Y, Rajasekhar VK, Ghajar CM, Matei I, Peinado H, Bromberg J, Lyden D. Tumour exosome integrins determine organotropic metastasis. Nature 2015 Nov 19;527(7578):329-35