Takuya IIDA
Professor, Bio-photophysics Group, Department of Physics, Graduate School of Science, Osaka Metropolitan University (OMU)
Director, Research Institute for Light-induced Acceleration System (RILACS), OMU
HP Link
Areas of Expertise
Bio-photophysics, Quantum Life Sciences, Nanophotonics
Research Overview
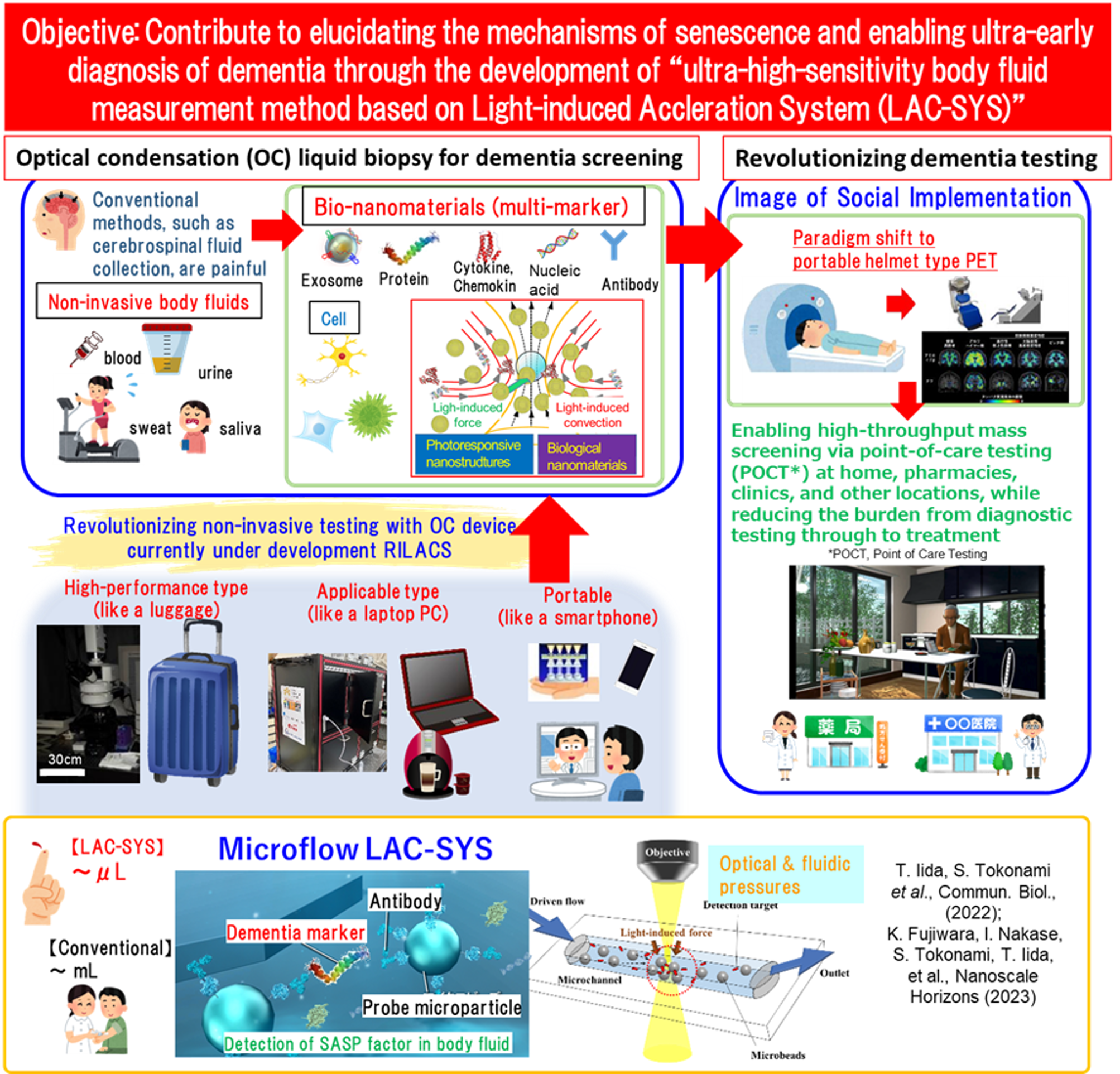
In this project, the “Optical Condensation Team” is responsible for developing ultra-high-sensitivity measurement technology for dementia markers using optical condensation (OC) method. We try to optimize the hardware of the OC testing system to enable ultra-high-sensitivity measurement of dementia markers in blood. Previously, Iida and colleagues have developed the “Light-Induced Acceleration System (LAC-SYS)”, which uses the synergistic effects of optical pressure and light-induced convection to optically condensate target substances and accelerate biochemical reactions such as antigen-antibody reactions. For example, by utilizing technologies such as the microflow LAC-SYS [Commun. Biol 2022], which enables the measurement of trace proteins at the attogram level (10-18g) from ultra-small liquid samples of several hundred nL within 3 to 5 minutes, and the nanobowl OC substrate [npj Biosensing 2024], which allows for simple antibody modification and highly sensitive, rapid measurement within 5 minutes, we will construct measurement systems for exosomes derived from aging cells—known as “Senescence-Associated Secretory Phenotype (SASP)” factors— cytokines, chemokines, and growth factors. Aiming to prevent diseases through cellular rejuvenation with drugs and the blocking of aging-promoting factors, we also develop core technologies— OC substrate and antibodies— collaborating with members of Osaka Metropolitan University (research collaborators: Shiho Tokonami, Ikuhiko Nakase, Taro Tachibana). We will contribute to this project by developing a widely accessible OC testing system that exhibits the high sensitivity and speed comparable with high-performance standard instruments, while providing affordable and easy-to-use results for measuring biomarkers in body fluids.
Main research contents
Measurement of Markers Derived from Senescent Cells Using the High-Performance Standard Model of the Light-Induced Acceleration System (LAC-SYS)
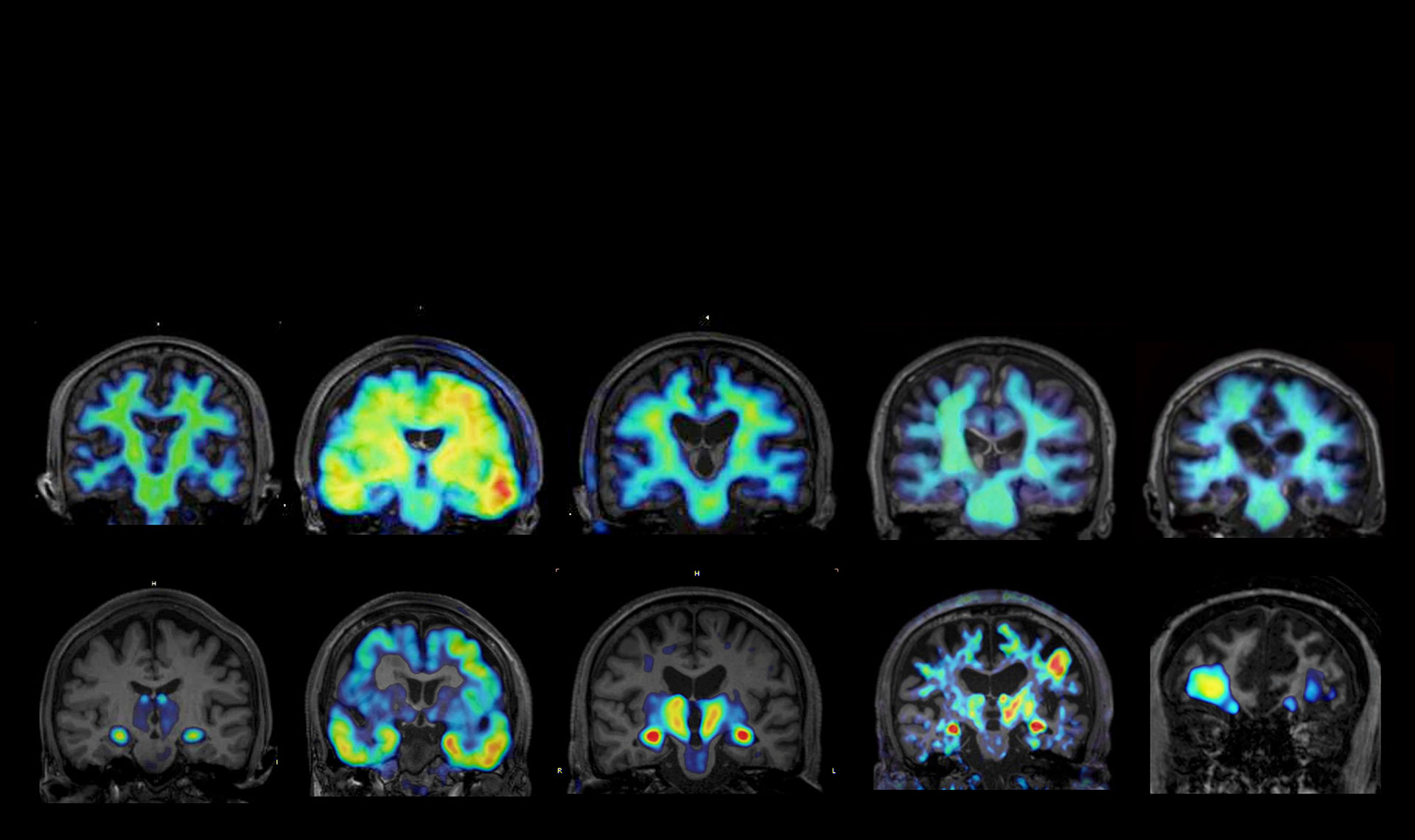
By exploiting the microflow LAC-SYS and nanobowl OC substrates developed to date, we will advance the development of assays targeting aggregative proteins, exosomes, and cytokines, enabling highly sensitive and rapid measurement of dementia markers such as aggregative proteins and SASP factors. Furthermore, we aim to establish an environment that allows for the simple analysis of components in supernatants from aging cells and in body fluids through comparison and integration with conventional analytical instruments (such as immunoassays). Additionally, we will develop and refine high-specificity, high-affinity antibodies for SASP factors optimized for the LAC-SYS. Furthermore, by integrating DX technologies such as AI, we aim to elucidate the correlation between measurement values in patient-derived body fluid samples—measured at multiple medical institutions—and the pathophysiology of dementia, focusing on comparisons with measurement results from PET and conventional immunoassays conducted by "Higuchi group" as the principal investigator. Through these efforts, we will contribute to the development of a platform for the ultra-early detection of dementia with high diagnostic accuracy.
Development of a Prototype LAC-SYS for Measuring SASP Factors in Body Fluids, and Its Implementation and Adoption in Medical Institutions
We will develop a widely applicable LAC-SYS based on a portable OC system that can be easily used in clinical settings, and proceed with the optimization of conditions for a light-induced immunoassay targeting SASP factors in body fluids. Specifically, we will optimize the optical and flow systems while correlating measurement data from the high-performance LAC-SYS with applicable LAC-SYS based on the portable OC system, and proceed with the development of a prototype capable of multi-marker measurement. In particular, we will proceed with the measurement of exosomes derived from senescent cells, as well as cytokines, chemokines, and growth factors derived from senescent cells, as well as the production of highly specific and high-affinity antibodies tailored to these targets, while incorporating feedback. We will construct an optical system capable of simultaneously measuring multiple samples with high reproducibility, and perform improvements and compositional analysis of the OC biochip. We will advance research and development aimed at measuring clinical samples in large-scale cohorts by installing the system at participating medical institutions, such as the Osaka Geroscience Center.
Major Achievements
- T. Iida*, “Development of Innovative Bio-Measurement Technology by Micro-Flow Light-Induced Acceleration”, READOUT Horiba Technical Report., 55, 11 (2021).
- T. Iida*, “Applications and prospects of light-induced acceleration systems for medical, food, and environmental measurements”, Oyo Buturi < Perspective >, 94 (5), 244 (2025).
- Shiho Tokonami*, Takuya Iida*, "Review: Novel Sensing Strategies for Bacterial Detection Based on Active and Passive Methods Driven by Ext Macroscopically Anisotropic Structures Produced by Light-induced Solvothermal Assembly of Porphyrin Dimers ernal Field", Analytica Chimica Acta, Vol.988, 1-16. (2017).
Selected Publications
- K. Hayashi, M. Tamura, M. Fujiwara, S. Tokonami*, T. Iida*, “Highly efficient three-dimensional optical condensation of nano- and micro-particles using a gold-coated optical fibre module”, Communications Physics, 9, 68 (2026).
- S. Toyouchi, S. Oomachi, R. Hasegawa, K. Hayashi, Y. Takagi, M. Tamura, S. Tokonami,* T. Iida*, “Single Nucleotide Polymorphism Highlighted via Heterogeneous Light-Induced Dissipative Structure”, ACS Sensors, 10, 751 (2025).
- M. Kanoda, K. Hayashi, Y. Takagi, M. Tamura, S. Tokonami*, T. Iida*, “High-throughput Light-induced Immunoassay with Milliwatt-level Laser under One-minute Optical Antibody-coating on Nanoparticle-imprinted Substrate”, npj Biosensing, 1, 1, (2024).
- K. Fujiwara, Y. Takagi, M. Tamura, M. Omura, K. Morimoto, I. Nakase*, S. Tokonami*, T. Iida*, “Ultrafast Sensitivity-Controlled and Specific Detection of Extracellular Vesicles Using Optical Force with Antibody-modified Microparticles in a Microflow System”, Nanoscale Horizons, 8, 1034-1042 (2023).
- I. Nakase*, M. Miyai, K. Noguchi, M. Tamura, Y. Yamamoto, Y. Nishimura, M. Omura, K. Hayashi, S. Futaki, S. Tokonami*, T. Iida*, “Light-Induced Condensation of Biofunctional Molecules around Targeted Living Cells to Accelerate Cytosolic Delivery”, Nano Letters, 22, 24, 9805–9814. (2022).
- T. Iida*, S. Hamatani, Y. Takagi, K. Fujiwara, M. Tamura, S. Tokonami, “Attogram-level light-induced antigen-antibody binding confined in microflow”, Communications Biology, 5, 1053 (2022).
- M. Tamura, T. Iida, K. Setoura*, “Plasmonic nanoscale temperature shaping on a single titanium nitride nanostructure”, Nanoscale, 14, 12589-12594 (2022).
- K. Hayashi, Y. Yamamoto, M. Tamura, S. Tokonami*, T. Iida*, “Damage-free Light-induced Assembly of Intestinal Bacteria with a Bubble-mimetic Substrate”, Communications Biology, 4, 385 (2021).
- S. Tokonami*, S. Kurita, R. Yoshikawa, K. Sakurai, T. Suehiro, Y. Yamamoto, M. Tamura, O. Karthaus, T. Iida*, “Light-induced assembly of living bacteria with honeycomb substrate”. Science Advances, 6, eaaz5757 (2020).
- M. Tamura*, T. Omatsu, S. Tokonami, T. Iida*, "Interparticle-interaction-mediated Anomalous Acceleration of Nanoparticles under Light-field with Coupled Orbital and Spin Angular Momentum", Nano Letters, 2019, 19, 8, 4873-4878 (2019).
- Y. Yamamoto, S. Tokonami*, T. Iida*, “Surfactant-Controlled Photothermal Assembly of Nanoparticles and Microparticles for Rapid Concentration Measurement of Microbes”, ACS Applied Bio Materials, 2, 1561–1568 (2019).
- M. Ueda, Y. Nishimura, M. Tamura, S. Ito, S. Tokonami*, T. Iida*, "Microflow-mediated optical assembly of nanoparticles with femtogram protein via shrinkage of light-induced bubbles", APL Photonics, 4, 010802 (2019).
- T. Iida*, Y. Nishimura, M. Tamura, K. Nishida, S. Ito, S. Tokonami*, “Submillimetre Network Formation by Light-induced Hybridization of Zeptomole-level DNA”, Scientific Reports, 6, 37768(1-9) (2016).
- Y. Nishimura, K. Nishida, Y. Yamamoto, S. Ito, S. Tokonami*, T. Iida*, “Control of Submillimeter Phase Transition by Collective Photothermal Effect”, J. Phys. Chem. C; 118, 18799-18804 (2014).