Shigeki KIYONAKA
Professor
Research Institute for Quantum and Chemical Innovation, Institutes of Innovation for Future Society
Department of Biomolecular Engineering, Graduate School of Engineering
Nagoya University
HP Link
Speciality
Chemical Biology
Research Overview
In Moonshot 7, entitled “Early Detection and Control of Dementia Onset Mechanisms Based on the Conceptual Framework Linking Glial Pathology to Senoinflammation,” I am responsible for the development of non-invasive, cell-type-specific technologies for the visualization and functional control of endogenous surface receptors in neurons and glial cells in living animals. Through this research, we aim to achieve a fundamental understanding of neurodegenerative diseases and to establish innovative intervention strategies grounded at the receptor level.
Recent advances in fluorescent protein technologies have made it routine to visualize the dynamics of intracellular proteins. However, most existing approaches rely on the overexpression of fusion proteins, which does not necessarily reflect physiological expression levels or native molecular behaviors. Because the expression and localization of intracellular proteins are tightly regulated, direct visualization of endogenous proteins is essential for accurately capturing their true dynamics under both physiological and pathological conditions. To date, we have developed visualization techniques targeting endogenous neurotransmitter receptors in neurons. In this project, we will further extend these approaches to establish high-precision in vivo visualization technologies for receptors endogenous to glial cells. This will enable us to capture cellular dysfunctions associated with neurodegenerative diseases, including dementia, with high temporal and spatial resolution.
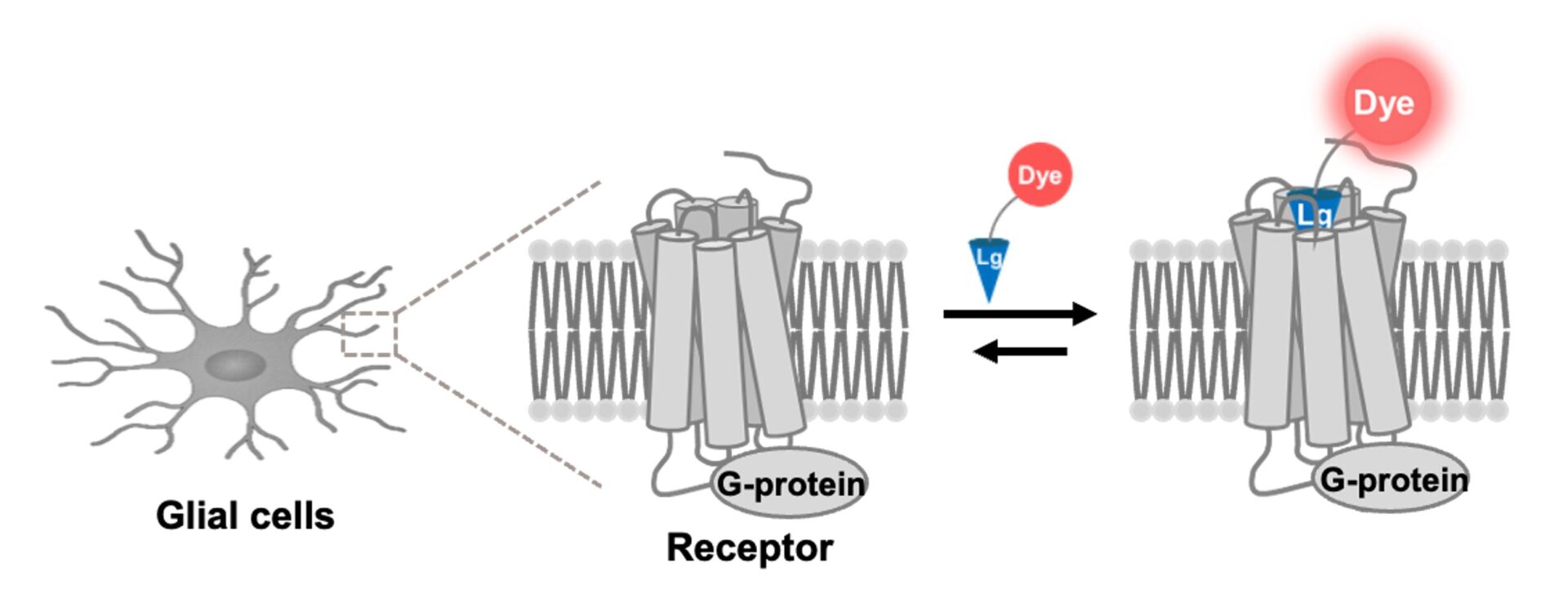
Optogenetic and chemogenetic approaches primarily enable artificial manipulation of neuronal activity and are not well suited for directly elucidating the functions of individual endogenous proteins. However, most existing methods artificially modulate neuronal activity and are not well suited for directly elucidating the functions of individual endogenous proteins. In this study, we will develop a novel receptor control technology that allows cell-type-specific, selective, and reversible inhibition of endogenous receptor activity at defined time points. This technology will not only advance our understanding of the physiological roles of receptors but also holds promise for next-generation drug discovery by enabling highly targeted interventions with reduced side effects.
Main research contents
Development of visualization probes to capture neuronal and glial alterations in neurodegenerative diseases
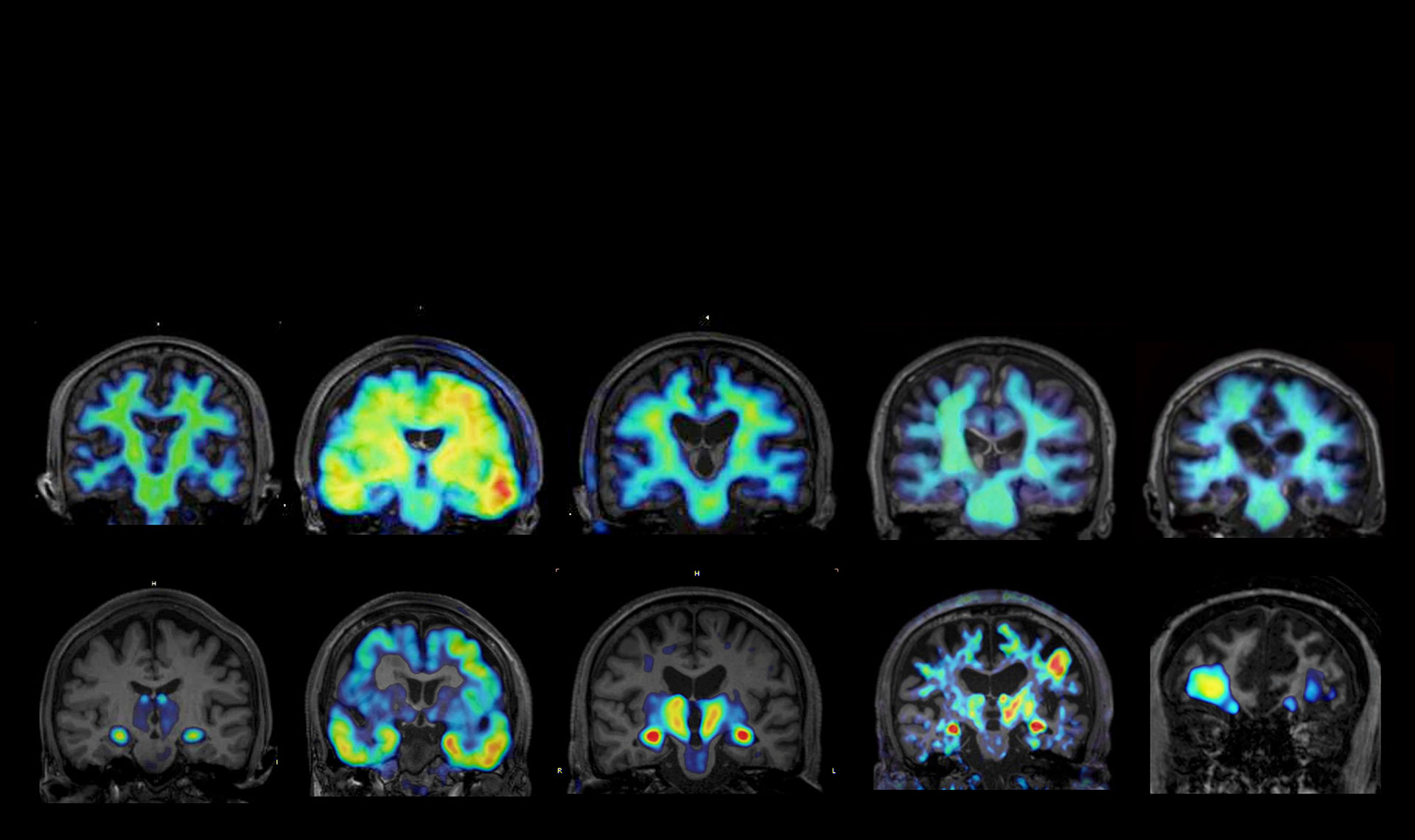
To date, we have developed visualization probes for AMPA-type glutamate receptors and have elucidated activity-dependent changes in synaptic AMPA receptor dynamics in cultured neurons. In the present study, we will position these probes as tools for capturing synaptic alterations associated with neurodegenerative diseases and further advance them toward in vivo visualization in the mouse brain through two-photon imaging, in collaboration with Prof. Hiroaki Wake at Nagoya University. In addition, we will develop probes to visualize alterations in glial cells. Specifically, we will design visualization probes targeting cell-surface marker proteins that are selective for individual glial cell types, such as microglia and astrocytes. Through in vivo imaging in collaboration with Prof. Wake, we aim to establish high-precision techniques for capturing the dynamics of glial cells in the living brain. Furthermore, we will develop high-sensitivity, high-resolution fluorescent probes optimized for two-photon imaging, enabling detailed analysis of receptor dynamics. We will also pursue the development of MRI probes targeting specific receptors, with the goal of establishing novel visualization technologies capable of capturing pathological alterations in deep brain regions at receptor-level resolution.
Elucidation of receptor functions in brain disorders through cell-type-specific interventional manipulation
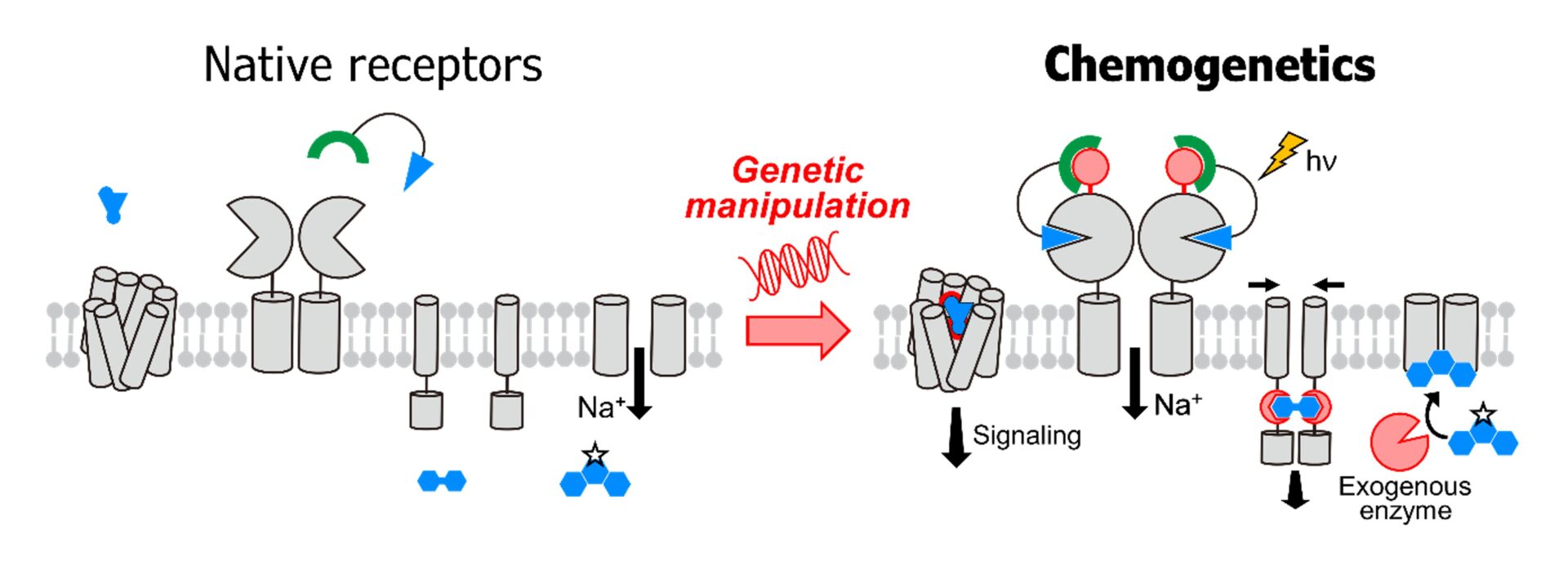
In this study, we aim to elucidate the roles of glial cells and their associated receptors in neurodegenerative diseases through cell-type-specific interventional manipulation. To date, we have developed an original approach that confers chemical controllability to receptors while preserving their native physiological functions. Unlike existing chemogenetic methods such as DREADDs, which manipulate overall cellular activity, our approach enables direct artificial control of the activity of endogenous receptors themselves. Whereas most conventional strategies have focused on the artificial activation of receptor signaling, this study will develop a novel chemogenetic technology that enables cell-type-specific and reversible inhibition of target receptor function by integrating genetic modification with chemical approaches. This strategy will allow precise suppression of the activity of specific receptors in defined cell types at arbitrarily chosen time points in living animals. By applying this method to models of brain disease, we will clarify the functional roles of receptors in distinct glial cell populations and, at the same time, explore its potential as a next-generation drug discovery platform that integrates genetic engineering and pharmacology.
Major Achievements
- Soga K., Fujiwara T., Nakagawa M., Shibata A., Adriel H., Yatsuzuka K., Kakegawa W., Yuzaki M., Hamachi I., Nango E., Kiyonaka S. Rapid and reversible fluorescent probe enables repeated snapshot imaging of AMPA receptors during synaptic plasticity. Sci. Adv., 11, eadt6683 (2025). DOI: 10.1126/sciadv.adt6683
- Suzuki H., Doura T., Matsuba Y., Matsuoka Y., Araya T., Asada H., Iwata S., Kiyonaka S. Photoresponsive adenosine derivatives for the optical control of adenosine A2A receptor in living cells. ACS Chem. Biol. 19, 2494-2501 (2024) DOI: 10.1021/acschembio.4c00583.
- Nonaka H., Sakamoto S., Shiraiwa K., Ishikawa M., Tamura T., Okuno K., Kondo T., Kiyonaka S., Susaki E.A., Shimizu C., Ueda H.R., Kakegawa W., Arai I., Yuzaki M., Hamachi I. Bioorthogonal chemical labeling of endogenous neurotransmitter receptors in living mouse brains. Proc. Natl. Acad. Sci. USA, 121, e2313887121 (2024) DOI: 10.1073/pnas.2313887121
- Ojima K., Kakegawa W., Yamasaki T., Miura Y., Itoh M., Michibata Y., Kubota R., Doura T., Miura E., Nonaka H., Mizuno S., Takahashi S., Yuzaki M., Hamachi I., Kiyonaka S. Coordination chemogenetics for activation of GPCR-type glutamate receptors in brain tissue. Nat. Commun., 13, 3167 (2022). DOI: 10.1038/s41467-022-30828-0
- Ojima K., Shiraiwa K., Soga K., Doura T., Takato M., Komatsu K., Yuzaki M., Hamachi I., Kiyonaka S. Ligand-directed two-step labeling to quantify neuronal glutamate receptor trafficking. Nat. Commun., 12, 831 (2021). DOI: 10.1038/s41467-021-21082-x.
- Wakayama S., Kiyonaka S., Arai I., Kakegawa W., Matsuda S., Ibata K., Nemoto L.Y., Kusumi A., Yuzaki M., Hamachi I. Chemical labeling for visualizing native AMPA receptors in live neurons. Nat. Commun., 8, 14850 (2017). DOI: 10.1038/ncomms14850.
- Kiyonaka S., Kubota R., Michibata Y., Sakakura M., Takahashi H., Numata T., Inoue R., Yuzaki M., Hamachi I. Allosteric activation of membrane-bound glutamate receptors using coordination chemistry within living cells. Nat. Chem., 8, 958-967 (2016). DOI: 10.1038/nchem.2554.