Message from the Project Manager
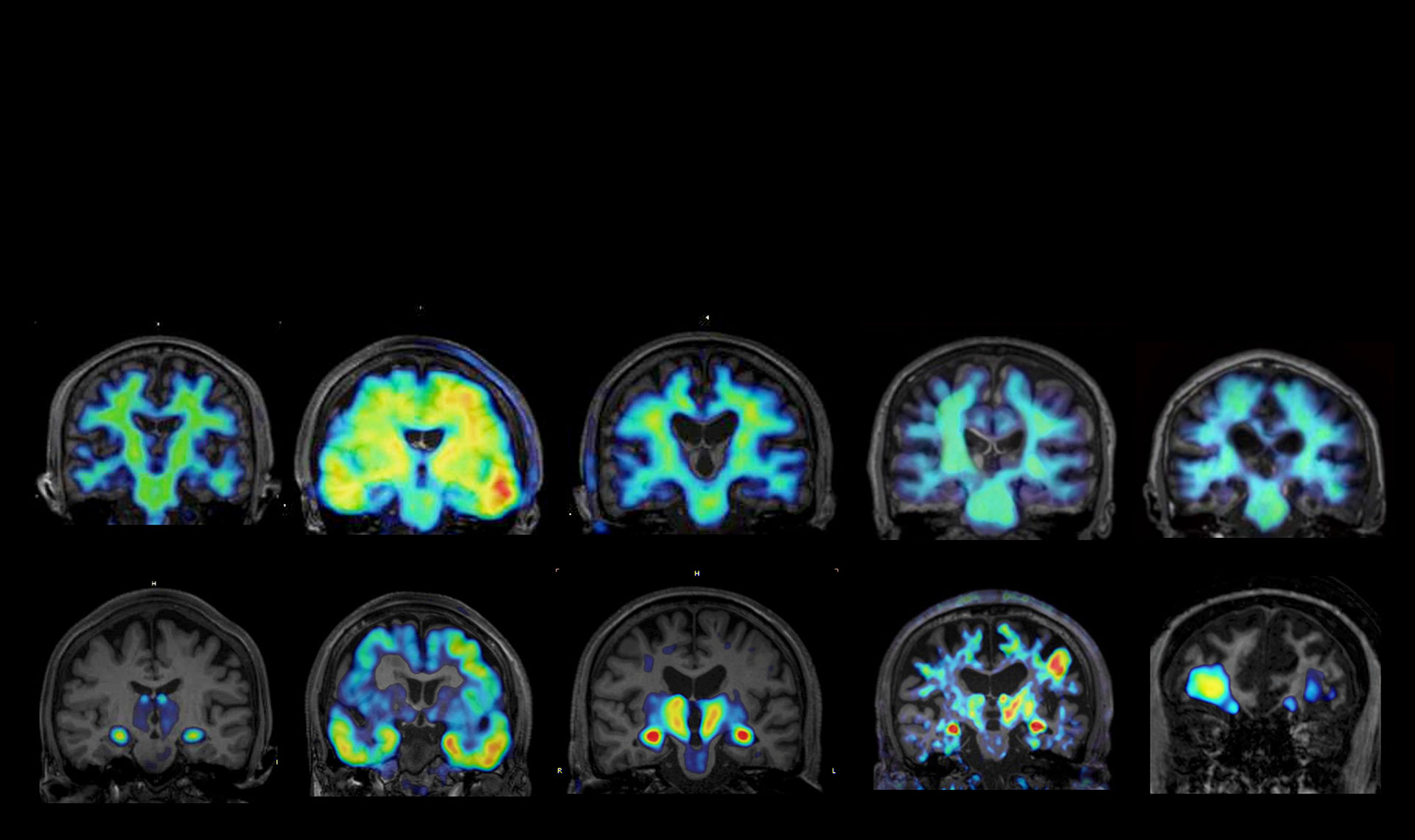
It is estimated that approximately 60 million people worldwide are currently living with dementia, with a similar number considered to be in the prodromal stage. Overcoming dementia is one of the most critical challenges facing humanity in realizing a happy and sustainable society of healthy longevity. A common pathological feature shared by many dementias, including Alzheimer’s disease, is the abnormal aggregation and deposition of proteins such as amyloid-β and tau in the brain. I have long been engaged in developing imaging and blood-based tests to visualize and detect these lesions.
In recent years, research and clinical implementation of therapies that target and remove pathological proteins have advanced rapidly through academia–industry collaboration, ushering in what could be called a new “golden age” of dementia diagnosis and treatment. In this Moonshot project, we position the concept of “brain senoinflammation,” which I have proposed, as a core framework for understanding dementia pathology. By detecting and correcting key molecular changes that occur in mid to late adulthood at a very early stage, we aim for a fundamental conquest of dementia. Leading researchers in brain inflammation, cellular senescence, protein aging, biometrology, and biorepository science have come together, with QST and the Osaka Geroscience Center (to be opened in May 2027) serving as the eastern and western hubs, respectively, to accelerate the social implementation of research accomplishments. With a strong conviction to realize a society in which people are freed from dementia and can live healthily and with peace of mind up to 100 years of age, we will fuse bold ideas with steady research efforts and turn dreams into reality.

Makoto Higuchi, MD, PhD
- Project Manager
- Director, Advanced Neuroimaging Center, Institute for Quantum Medical Science,
- National Institutes for Quantum Science and Technology (QST)
- Professor, Neuroe tiology and Diagnostic Science, Graduate School of Medicine, Osaka Metropolitan University
What Is the
“Brain Senoinflammation” Project?
Early detection and modulation of the dementia pathogenesis based on the concept
evolving from glial pathology to senoinflammation
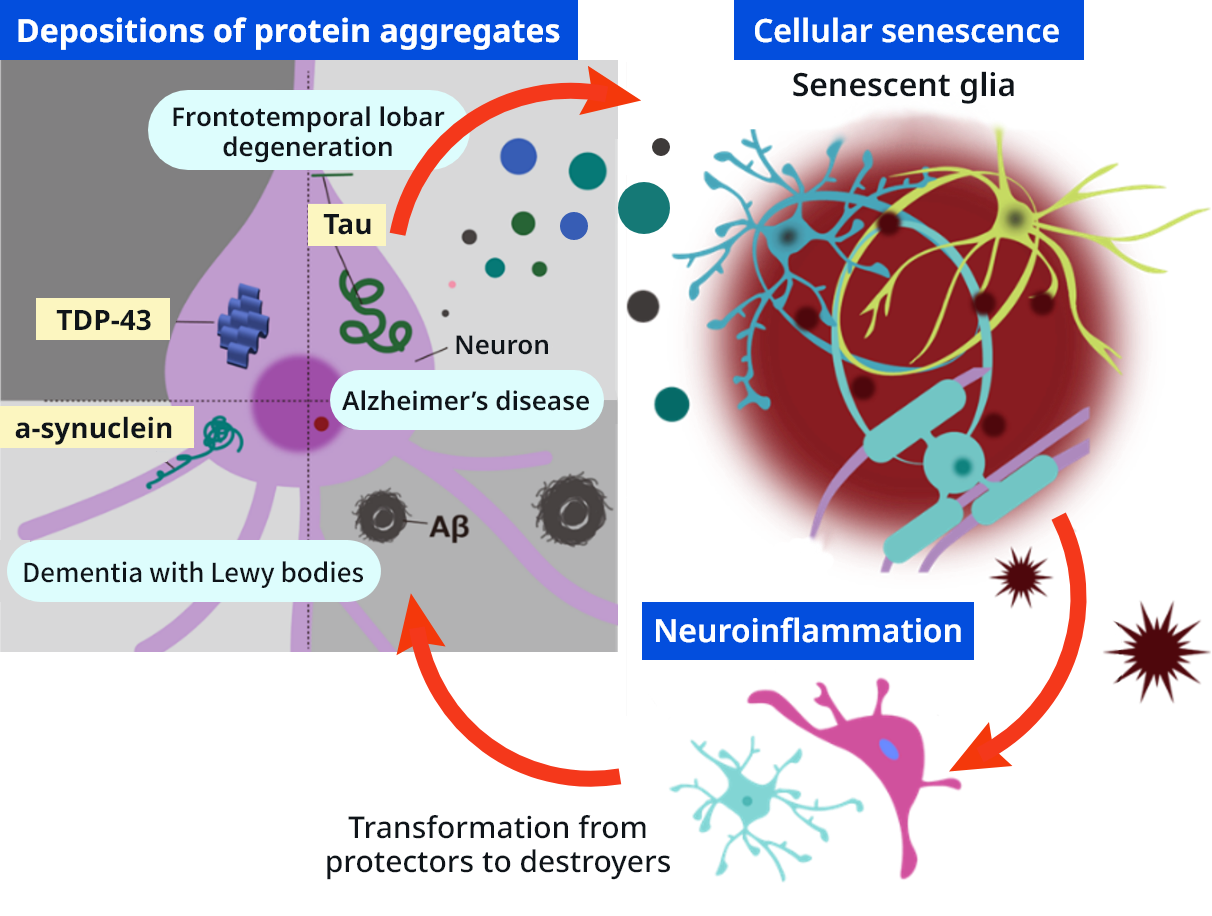
Dementia is thought to be caused mainly by three harmful changes in the brain: (1) abnormal accumulation of proteins such as amyloid-β and tau as fibrillar “waste” (brain protein aging), (2) inflammatory activation of glial cells that normally protect the brain, and (3) senescence of neurons and glial cells. In addition to the mutual amplification between cellular senescence and inflammation termed “senoinflammation”, the addition of brain protein aging creates a triadic interaction, which we refer to as “brain senoinflammation,” and which represents the essence of dementia pathology.
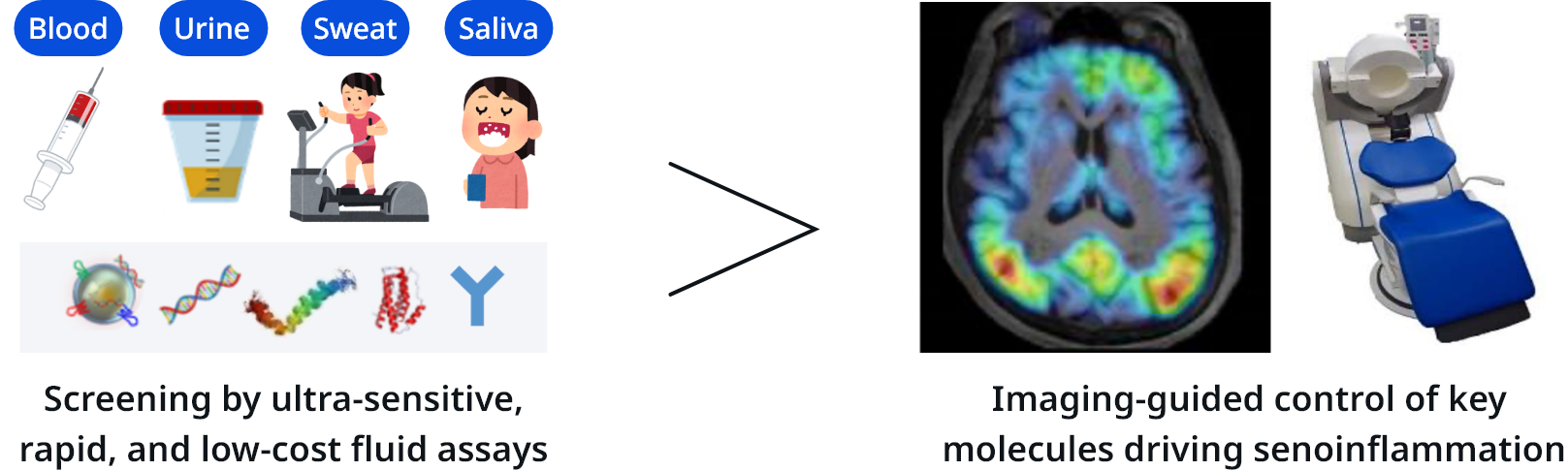
This project aims to identify and control the key molecules that drive brain senoinflammation. By targeting these key molecules, we will promote miniaturization and high-efficiency development of blood-based and imaging diagnostics, as well as the creation of novel therapeutics, with the ultimate goal of realizing a society of healthy longevity in which dementia is overcome.
Brain senoinflammation
Next-generation diagnostic & therapeutic workflow
Key objectives
-
Identification and evaluation of key molecules
Identify the most effective targets for overcoming dementia and evaluate pathology using blood tests and imaging.
-
Integrated diagnosis and therapy
Intervene while visualizing key molecules and verify the efficacy of drugs and next-generation supplements.
-
Rapid detection and implementation
Enable simple detection from small samples and promote social implementation of evidence-based technologies.
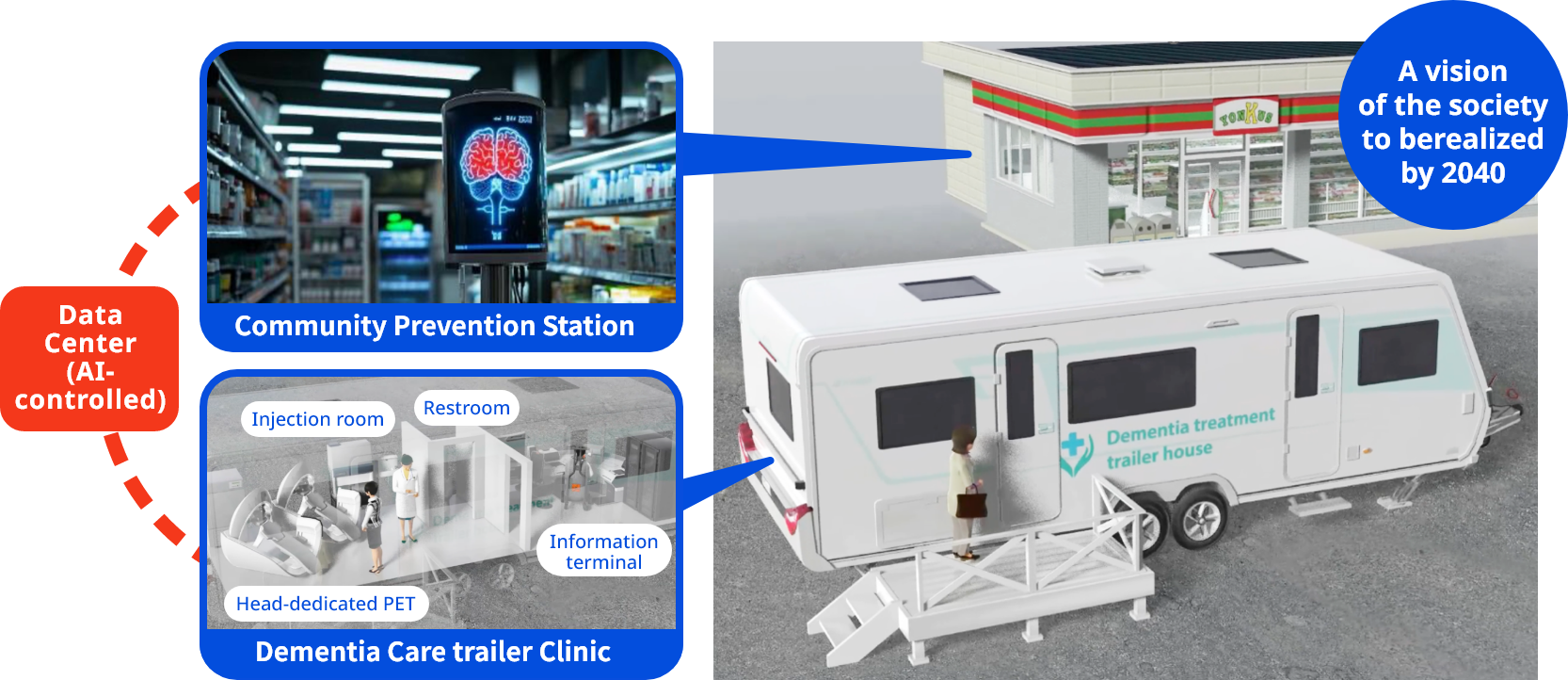
Conceptual image of the society to be realized by the “Brain Senoinflammation” project
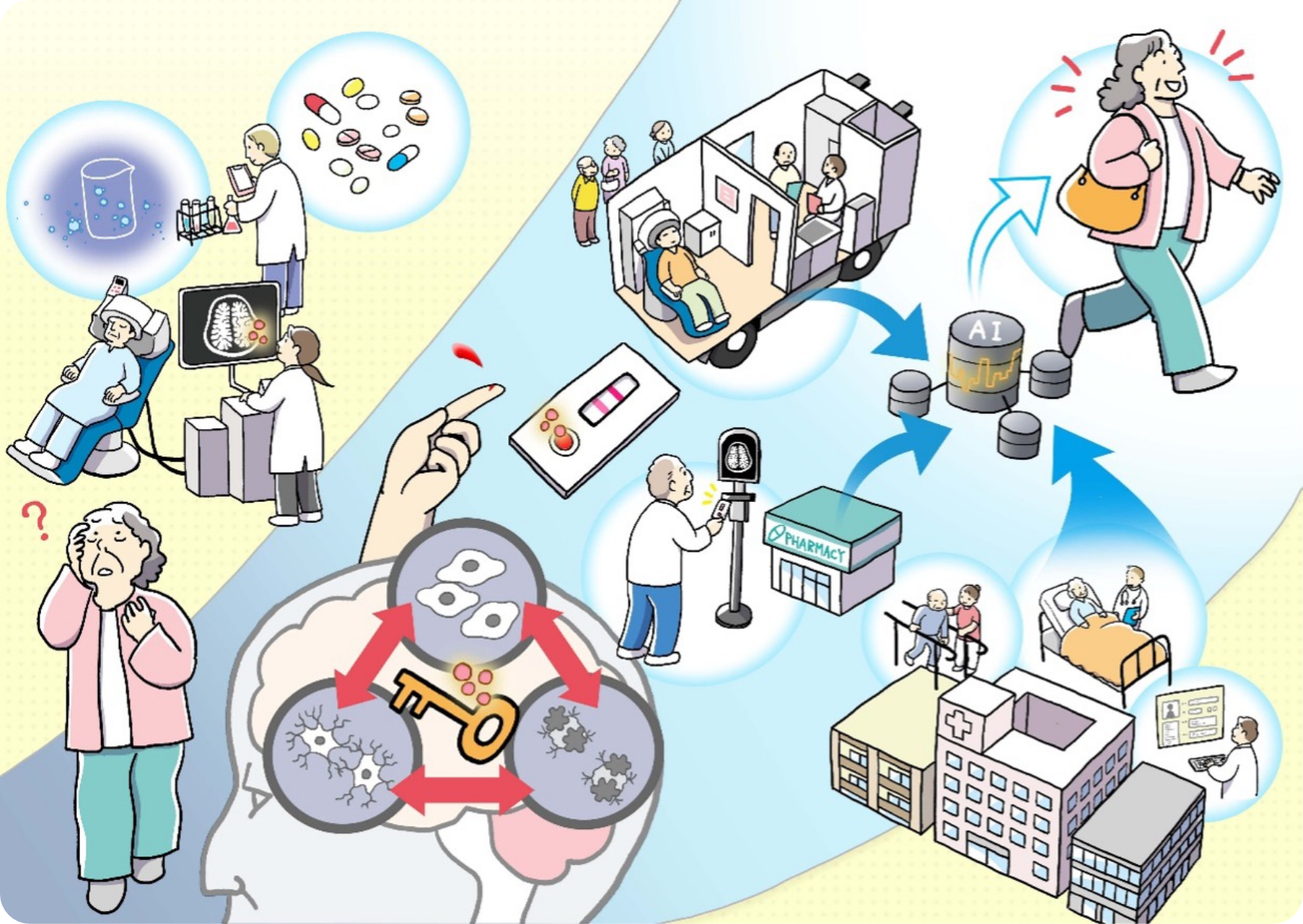
Illustration of “anywhere diagnosis, anywhere treatment” for dementia pursued by the “Brain Senoinflammation” project